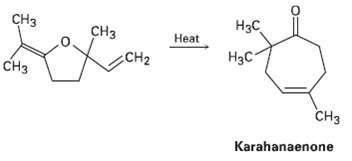
Karahanaenone, a terpenoid isolated from oil of hops, has been synthesized by the thermal reaction shown. Identify
Question:
Karahanaenone, a terpenoid isolated from oil of hops, has been synthesized by the thermal reaction shown. Identify the kind of pericyclic reaction, and explain how karahanaenone isformed.
Transcribed Image Text:
CHз Нас CHз Нeat H3C CH2 CHз снз Karahanaenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
HC HC 01 CH3 33 shift heat H...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Safrole, a substance isolated from oil of sassafras, is used as a perfumery agent. Propose a synthesis of safrole from catechol (1,2-benzenediol). CH2CH=CH2 Safrole
-
The sex hormone estrone has been synthesized by a route that involves the following step. Identify the pericyclic reactions involved, and propose amechanism. CH CH Heat |CH30 CH0 Estrone methyl ether
-
A South Korean research group has isolated and synthesized "daumone," the pheromone that induces hibernation in Caenorhabditis elegans worms when food becomes scarce, thus extending their life span...
-
Warnerwoods Company uses a periodic inventory system. It entered into the following purchases and sales transactions for March. Date Activities Units Acquired at Cost Units Sold at Retail Mar. 1...
-
Speculate as to why Microsoft used cash rather than some other form of payment to acquire Nokia?
-
Explain why managers may prefer to expense outlays on intangibles rather than capitalise them.
-
A gasoline engine consumes 8 litres of fuel per hr. having specific gravity of 0.75 while it develops \(25 \mathrm{~kW}\) power. If the C.V. of fuel used is \(44200 \mathrm{~kJ} / \mathrm{kg}\),...
-
The following remark was overheard at a meeting between managers and a systems analysis team at the Fence-Me-In fencing company: You told us the prototype would be finished three weeks ago. Were...
-
I believe that an increase in the use of robotics will cause both a loss of jobs and a shifting of jobs. Unfortunately, I belive that the loss of jobs will be a greater number than the amount of jobs...
-
Euro Corporation, a U.S. corporation, operates through a branch in Germany. During 2022, the branch reported taxable income of $1,000,000 and paid German income taxes of $300,000. In addition, Euro...
-
The following synthesis of dienones occurs readily. Propose a mechanism to account for the results, and identify the kind of pericyclic reactioninvolved. Heat Acid catalyst
-
The 1H NMR spectrum of bullvalene at 100 C consists only of a single peak at 4.22 ?. Explain. Bullvalene
-
Why are IT job prospects so strong?
-
The impulse responses of four linear-phase FIR filters h [n], h [n], h3 [n], and h [n] are given below. h [n] = -6[n] -0.58[n 1] +8[n-2] - 8[n-4] +0.58[n - 5] + [n-6] h [n] = 0.48 [n] -0.38 [n 1]...
-
Algo Rhythm Ltd (ARL) receives fixed rate income of 4% p.a. paid semi-annually, but has borrowed by issuing floating rate notes with a total face value of $3 million. The floating rate notes mature...
-
You may need to use the appropriate technology to answer this question. Three admission test preparation programs are being evaluated. Suppose the scores obtained by a sample of 20 people who used...
-
Crane Corp. is thinking about opening a soccer camp in southern California. To start the camp, Crane would need to purchase land and build four soccer fields and a sleeping and dining facility to...
-
You are provided with the trial balance of Twirl Ltd. as at 31st December 2019: Bank 78,000 Ordinary shares at 1 each 216,000 Retained earnings at 1st January 2019 476,400 Share premium 26,160...
-
Describe your ideal organizational culture using the seven dimensions of organ-izational culture. What constraints would this culture put on organizational managers behavior? How? Does it constrain...
-
Juarez worked for Westarz Homes at construction sites for five years. Bever was a superintendent at construction sites, supervising subcontractors and moving trash from sites to landfills. He...
-
Critics of high-frequency trading often complain that the practice is unfair because most investors do not have the technical ability to compete with the larger, betterfinanced institutions. For...
-
The alkane formed by hydrogenation of (S)-4-methyl-1-hexyne is optically active, but the one formed by hydrogenation of (S)-3-methyl-1-pentyne is not. Explain. Would you expect the products of...
-
How do bond distances and bond strengths change with electronegativity in the series NH3, H2O, and HF?
-
All the following reactions have been described in the chemical literature and proceed in good yield. In some cases the reactants are more complicated than those we have so far encountered....
-
Because of its high dielectric strength, SF6 (sulfur hexafluo-ride) gas is widely used as an insulator and a dielectric in HV applications such as HV transform-ers, switches, circuit breakers,...
-
An eagle is flying horizontally at a speed of 2.9 m/s when the fish in her talons wiggles loose and falls into the lake 4.4 m below. Calculate the magnitude of the velocity of the fish relative to...
-
A 5.0 g bullet moving 325 m/s is shot into a 1.25 kg block, which slides for 1.35 seconds across the surface it is on before coming to rest. What is the average force of kinetic friction between the...

Study smarter with the SolutionInn App


