Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of
Question:
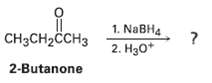
Ketones undergo a reduction when treated with sodium borohydride, NaBH4. What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm?1 arid M+ = 74 in the mass spectrum?
Transcribed Image Text:
1. NABH4 2. Hзо* |CH3CH2CCHЗ 2-Butanone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
CH3CH2CCH3 1 NaBH4 2 H3O OH ...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is the structure of ATCase?
-
What is the structure of the global beer industry?
-
What is the structure of the beer industry?
-
What do you think people would say about Corrie from the few quotes we have from her book? What was her personality like? Do you think she handled her incarceration differently than Elie Wiesel?...
-
Industries with high barriers to entry often have high barriers to exit. Explain.
-
A 4.00-g sample of a mixture of CaO and BaO is placed in a 1.00-L vessel containing CO2 gas at a pressure of 730 torr and a temperature of 25oC. The CO2 reacts with the CaO and BaO, forming CaCO3 and...
-
Why is it important for an organization to be flexible when developing information systems?
-
Using the appropriate tables, solve each of the following: Required 1. Beginning December 31, 2008, five equal withdrawals are to be made. Determine the equal annual withdrawals if $30,000 is...
-
In 1985, 29 year old Anthony Ray Hinton was found guilty on two counts of capital murder and sentenced to death by electric chair. He spent nearly 30 years on death row for crimes he did not commit....
-
Brookland Insurance Company (BIC) has a QST with Cypruss Re. The treaty has a $250,000 limit, and a retention of 25%. The following three policies are issued by BIC and are subject to the pro-rata...
-
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C 7 H 14 O. What is the...
-
Nitriles, RC N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propane nitrile, CH3CH2C N, if it has IR absorptions at...
-
Use synthetic division to find the quotient and remainder when: -4x 3 + 2x 2 x + 1 is divided by x + 2
-
The following transactions occurred during 2 0 2 4 for the Beehive Honey Corporation: February 1 Borrowed $ 1 9 , 0 0 0 from a bank and signed a note. Principal and interest at 1 2 % will be paid on...
-
Find the zeros of the following polynomial. f(x)=x3-8x2-x+8 List the zeroes separated by commas. For example, if you found that the zeroes were x=
-
rom the Stern vs. Lucy Webb Hayes case: What was the core issue? What are the key duties or responsibilities of a governing board? Would or could this happen today? Why or why not? What measures...
-
Why is oil connected to up to half of all wars between countries? Why do you think we are more likely to launch large, sweeping national security initiatives after events such as wars and terrorist...
-
Stop and frisk is a controversial topic, explain whether you believe if stop and frisk is a form of harassment or is stop and frisk necessary for law enforcement. In your answer explain what are the...
-
You keep a chalkboard eraser pressed against the chalkboard by using your finger to exert a horizontal force on the back of the eraser. (a) Which type of force (call it force A) keeps the eraser from...
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
The solubility of carbon tetrachloride (CCl 4 ) in water at 25 C is 1.2 g/L. The solubility of chloroform (CHCl 3 ) at the same temperature is 10.1 g/L. Why is chloroform almost ten times more...
-
Predict the products of the reactions of the following compounds with chromic acid and also with PCC. (a) Cyclohexanol (b) 1-methylcyclohexanol (c) Cyclopentylmethanol (d) Cyclohexanone (e)...
-
Two products are observed in the following reaction. (a) Suggest a mechanism to explain how these two products are formed. (b) Your mechanism for part (a) should be different from the usual mechanism...
-
Give the structures of the products you would expect when each alcohol reacts with (1) HCI, ZnCI2; HBr; (3) PBr3: (4) P/I2; (5) SOCI2- (a) butan-1-ol (b) 2-methylbutan-2-ol (c) 2,2-dimethylbutan-1-ol...
-
Discuss the automotive industry in Munich Germany to include: a) Current size of the industry in terms of revenues, b) Detailed information on BMW in Munich, Baravia, c) BMWs main competitors in...
-
Using the Internet, identify a known sexual offender who has been arrested, convicted, and sentenced. Using the available information, describe each of the following: (a) The type of behavior that...
-
Of our secondary and tertiary education system, are we doing a great job preparing students for the professional world post-college, or should that be placed upon the employer? If we need have a...

Study smarter with the SolutionInn App


