Look at the five resonance structures for Phenanthrene (Problem 15.26) and predict which of its carboncarbon bonds
Question:
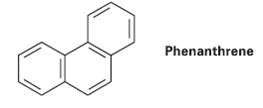
Look at the five resonance structures for Phenanthrene (Problem 15.26) and predict which of its carbon—carbon bonds is shortest.
Transcribed Image Text:
Phenanthrene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
The circled bond is represented as a doubl...View the full answer

Answered By

Ayush Jain
Subjects in which i am expert:
Computer Science :All subjects (Eg. Networking,Database ,Operating System,Information Security,)
Programming : C. C++, Python, Java, Machine Learning,Php
Android App Development, Xamarin, VS app development
Essay Writing
Research Paper
History, Management Subjects
Mathematics :Till Graduate Level
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The resonance structures of carbon monoxide are shown below. Show how each structure can be converted into the other using the curved-arrow notation. :C-: :C=0:
-
Phenanthrene has five total resonance structures. One is shown here. Draw the other four. Which carbon-carbon bond of Phenanthrene would your predict to be the shortest? Phenanthrene
-
For each set of resonance structures that follow, add a curved arrow that shows how electrons in the left formula shift to become the right formula, and designate the formula that would contribute...
-
Two franchising experts recently debated the issue of whether new college graduates should consider franchising as a pathway to entrepreneurship. Jeff Elgin said recent college graduates are not...
-
Discuss the relative merits of internal versus external recruitment. Give an example of a situation in which each of these approaches might be particularly effective.
-
Determine the approximate density of a high-leaded brass that has a composition of 64.5 wt% Cu, 33.5 wt% Zn, and 2.0 wt% Pb.
-
In an experiment designed to determine the specific heat ratio \(\gamma\) for a certain gas, measurements of the volume and corresponding pressure \(p\) produced the data: Assuming the ideal gas law...
-
The following refers to units processed in Sunflower Printings binding department in March. Prepare a physical flow reconciliation. Units of Percent of Product Conversion Added Beginning work in...
-
The following situation is an example for a fire detection and suppression system in an office building. The Initiating Event (IE) for the Event Tree is "fire starts", with a frequency of once every...
-
Complete the payroll register for this pay period and update the Employee Earnings Record form for each employee with the corresponding information. The Step-2 of Form W-4 is unchecked. The amount...
-
There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour. Phenanthrene
-
In 1932, A. A. Levine and A. G. Cole studied the ozonolysis of o-xylene and isolated three products: glyoxal, 2, 3-butanedione, and pyruvaldehyde: In what ratio would you expect the three products to...
-
A firm has earnings before interest and tax of $1,000,000, interest of $200,000, and net income of $400,000 in Year 1. Required a. Calculate the degree of financial leverage in base Year 1. b. If...
-
Juett Company produces a single product. The cost of producing and selling a single unit of this product at the company's normal activity level of 70,000 units per month is as follows: Direct...
-
In building a storage shed, workers use nails from a large bin. The bin is suspended from a vertical spring twice during the day to measure the usage of nails. At the beginning of the day, the spring...
-
Given a demand and supply curve as follows: P = -3Q+10 P = Q+4 Determine the change in equilibrium quantity if there is a change in the value of c of 0.2 and how about the graph changes . demand and...
-
A gift of land valued at $80,000 was given to Dorinda by her long-time friend, Justine.The land's original cost to Justine was $30,000. As a result of the transfer, Justine paid a gift tax of...
-
Explain how a company could (a) improve its credit rating withsuppliers after paying some late; (b) lower its cost of financing when the market interest rate has increased. (financial magmt)
-
You borrow \($5\),000 at 10 percent per year and will pay off the loan in three equal annual payments starting 1 year after the loan is made. The end-of-year payments are \($2\),010.57. Which of the...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
What is the significance of the equilibrium constant? What does a large equilibrium constant tell us about a reaction? A small one?
-
Give complete mechanisms for the formation of Z-Ala, its activation by ethyl chloroformate, and the coupling with valine.
-
Show how you would synthesize Ala-Val-Phe-Gly-Leu starting with Z-Ala-Val-Phe.
-
Show how the solution-phase synthesis would be used to synthesize Ile-Gly-Asn.
-
Assume that a robot systems constant failure and repair rates are 0.0004 failures per hour and 0.002 repairs per hour, respectively. Calculate the robot system steady-state unavailability and...
-
March 6, 2018 David Beckham's Latest Pitch: Men's Cosmetics. By Robert Williams Men of the world, David Beckham has a message for you: Moisturize. "It's the biggest thing," he says in a London...
-
3. On January 1, an investment fund was opened with an initial balance of 5000. Just after the balance grew to 5200 on July 1, an additional 2600 was deposited. The annual effective yield rate for...

Study smarter with the SolutionInn App


