There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour. Phenanthrene
Question:
There are five resonance structures of Phenanthrene, one of which is shown. Draw the otherfour.
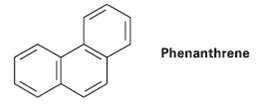
Transcribed Image Text:
Phenanthrene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The circled bond is represented as a doubl...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
There are two contributing resonance structures for an anion called acetaldehyde enolate, whose condensed molecular formula is CH2CHO-. Draw the two resonance contributors and the resonance hybrid,...
-
Draw contributing resonance structures and a hybrid resonance structure that explain two related facts: the carbon-oxygen bond distances in the acetate ion are the same, and the oxygen's of the...
-
Draw two resonance structures for diazomethane, CH2N2. Show formal charges. The skeletal structure of the molecule is C N N
-
Based on your reading of the rules of Professional Code of Conduct as given by AICPA, which part of the code did the accountants of Enron, Waste Management or Wells Fargo have violated and how about...
-
What pros and cons of downsizing do you think apply to this example?
-
Calculate the fraction of atom sites that are vacant for lead at its melting temperature of 327C (600 K). Assume an energy for vacancy formation of 0.55 eV/atom.
-
With reference to Exercise 11.75, use the method of Section 11.2 to construct a \(95 \%\) confidence interval for \(\gamma\). State what assumptions will have to be made. Data From Exercise 11.75...
-
The current sections of Sunn Inc.'s balance sheets at December 31, 2016 and 2017 are presented here. Sunn's net income for 2017 was $153,000. Depreciation expense was $27,000. Instructions Prepare...
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
In 1738, J. Bernoulli investigated the St. Petersburg paradox, which works as follows. You have the opportunity to play a game in which a fair coin is tossed repeatedly until it conies up heads. If...
-
There are four resonance structures for anthracene, one of which is shown. Draw the otherthree. Anthracene
-
Look at the five resonance structures for Phenanthrene (Problem 15.26) and predict which of its carboncarbon bonds is shortest. Phenanthrene
-
If individual accountability is an essential element of professionalism, why is it also important to be an effective team player? Explain your answer.
-
You are starting a business selling products A and B. The making of one product A requires 60 minutes of labor and its materials that cost P21. The making of one product B requires 35 minutes of...
-
In the context of trading and risk systems quantitative development: A) What are low level and high-level programming? Explain and provide at least two examples of each. B) Explain the three layers...
-
On the day she retired, Kate had $713,300 in retirement savings. She expects to earn 4.5 percent, compounded monthly , and live 24 more years. How much can she withdraw from her savings each month...
-
There is a portfolio of 7 possible investments to choose from, the net present value (NPV), of the profit of each of the possible investments as well as the capital that you have to invest per year...
-
A 1 liter bag of NS with 20meq of KCL is to infuse over 8 hours. At 4 hours you notice there is 400ml remianing in the bag. to complete the infusion on time, what should the pump be set to
-
One of your financing options is a balloon loan on a \($200\),000 thirty-year mortgage with monthly payments and an interest rate of 7.5 percent per year. Which of the following statements is...
-
Identify the most stable compound:
-
The reaction shown here has a K p = 4.5 * 10 2 at 825 K. Find K c for the reaction at this temperature. CH(g) + CO(g) 2 CO(g) + 2 H(g)
-
The Sanger method for N-terminus determination is a less common alternative to the Edman degradation. In the Sanger method, the peptide is treated with the Sanger reagent, 2, 4-dinitrofluorobenzene,...
-
Show where trypsin and chymotrypsin would cleave the following peptide. Tyr-Ile-Gln-Arg-Leu-Gly-Phe-Lys-Asn-Trp-Phe-Gly-Ala-Lys-Gly-Gln-Gln NH2
-
After treatment with peroxyformic acid, the peptide hormone vasopressin is partially hydrolyzed. The following fragments are recovered. Propose a structure for vasopressin. Phe-Gln-Asn...
-
Part 1 - Rent Calculation Assume the following for a floor in a multistory office building: total usable area: 62,500 sq ft; Total common area: 4,100 sq ft; total rentable area: 66,600 sq ft; Tenant...
-
14. Consider the following information on three stocks: Probability of State Rate of Return if State Occurs State of Economy of Economy Stock A Stock B Stock C Boom 0.45 0.55 0.35 0.65 Normal 0.50...
-
B2 ) Consider the following information on a portfolio of three stocks: State of Economy Probability of State of Economy Boom .15 Stock A Rate of Return .05 Stock B. Rate of Return 21 Stock C Rate of...

Study smarter with the SolutionInn App


