M.-G. Olivier and R. Jadot Cl. Chem. Eng. Data 42,230 (1997)) studied the adsorption of butane on
Question:
M.-G. Olivier and R. Jadot Cl. Chem. Eng. Data 42,230 (1997)) studied the adsorption of butane on silica gel. They report the following amounts of absorption (in moles per kilogram of silica gel) at 303 K:
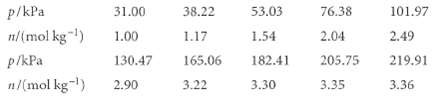
Fit these data to a Langmuir isotherm, and determine the value of n that corresponds to complete coverage and the constant K.
Transcribed Image Text:
p/kPa n/(mol kg-¹) p/kPa n/(mol kg-¹) 31.00 1.00 130.47 2.90 38.22 1.17 165.06 3.22 53.03 1.54 182.41 3.30 76.38 2.04 205.75 3.35 101.97 2.49 219.91 3.36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The Langmuir isotherm is Kp 1 Kp 8 P P n no Knx pkPa nmol ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physical Chemistry questions
-
The adsorption of a gas is described by the Langmuir isotherm with K = 0.777 kPa-1 at 25C. Calculate the pressure at which the fractional surface coverage is (a) 0.20, (b) 0.75.
-
Olivier Corporation reported the following pre-tax amounts for the year ended August 31, 2017: profit before income tax (on the company's continuing operations), $320,000; loss from operations of...
-
In a series RLC circuit, R = XC = CL = 40- for a particular driving frequency. (a) This circuit is (1) inductive, (2) capacitive, (3) in resonance. Explain your reasoning. (b) If the driving...
-
On March 19, Modern Kitchens, a retail store, received Credit Memorandum 244 for $4,290 from J & M Appliance Corporation. The credit memorandum covered a return of damaged trash compactors originally...
-
For an iron-carbon alloy of composition 3 wt% C-97 wt% Fe, make schematic sketches of the microstructure that would be observed for conditions of very slow cooling at the following temperatures:...
-
If permitted, a lessee generally prefers to account for leases using the operating leases method for financial reporting and the capital lease method for tax reporting. Explain.
-
Tip Top Advertising, Inc., engaged in the following business transactions during July of 2010: Tip Top Advertising, Inc., uses the following accounts: Cash, Accounts Receivable, Supplies, Building,...
-
Lululemon athletica, a high-end retail chain dedicated to yoga and fitness apparel, recently experienced several years of record growth. Founded and headquartered in Vancouver, British Columbia,...
-
Prepare journal entries to record the following transactions for a corporation for the current year. January 3 Issued 33,600 shares of $2 par value common stock at $22 cash per share. Declared cash...
-
For the past several years, Jeff Horton has operated a part-time consulting business from his home. As of April 1, 2016, Jeff decided to move to rented quarters and to operate the business, which was...
-
A. Akgerman and M. Zardkoohi (J. Chem. Eng. Data 41,185 (1996)) examined the adsorption of phenol from aqueous solution on to fly ash at 20e. They fitted their observations to a Freundlich isotherm...
-
In an experiment on the Pt IH,I H+ electrode in dilute H,S04 the following current densities were observed at 25e. Evaluate ET and jo for the electrode. How would the current density at this...
-
Let \(B\) be a Brownian motion and \[\begin{aligned}T_{a}^{(u)} & =\inf \left\{t: B_{t}+u t=a ight\} \\G_{a}^{(u)} & =\sup \left\{t: B_{t}+u t=a ight\}\end{aligned}\] Prove that \[\left(T_{a}^{(u)},...
-
What lessons can be drawn from historical instances of social change, and how can these lessons be applied to contemporary movements to facilitate more effective and sustainable societal...
-
A tennis player hits a ball 1.9 m above the ground. The ball leaves his racquet with a speed of 14.4 m/s at an angle 5.5 above the horizontal. The horizontal distance to the net is 8 m, and the net...
-
Arnold Vimka is a venture capitalist facing two alternative investment opportunities. He intends to invest $1,000,000 in a start-up firm. He is nervous, however, about future economic volatility. He...
-
How do cultural artifacts such as media, art, and literature reflect and influence trends in social change, and what is their role in shaping public consciousness around emerging social issues ?
-
Figure 1 shows a diagram of your client's network showing an enterprise network design. Your client which is a timber wholesaler in Korea has four servers accessible via the Internet and...
-
The bias voltages of the circuit shown in Figure P7.67 are changed to \(V^{+}=3 \mathrm{~V}\) and \(V^{-}=-3 \mathrm{~V}\). The input resistances are \(R_{i}=4 \mathrm{k} \Omega\) and \(R_{G}=200...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Use Newtons method to find all roots of the equation correct to six decimal places. 1/x = 1 + x 3
-
Carbonate and hydrogen carbonate (bicarbonate) ions contribute to buffering in a variety of natural systems. You are investigating their role in groundwater percolating through limestone hills into a...
-
Predict the standard potential of each of the following galvanic cells: 3+ (a) Pt(s) Cr+ (aq), Cr+ (aq)||Cu+ (aq) |Cu(s) (b) Ag(s) AgI(s) I (aq)||CI (aq)|AgCl(s) Ag(s) (c) Hg(1) HgCl (s) CIT(aq) |Hg+...
-
Estimate the enthalpy of deprotonation of formic acid at 25C, given that K a = 1.765 * 10 4 at 20C and 1.768 * 10 4 at 30C.
-
Explain and contrast unique ( individual , unsystematic ) risk of a firm and market ( systematic ) risk. Explain how unique risk and market risk can be measured.
-
Carmen Camry operates a consulting firm called Help Today, which began operations on December 1. On December 31, the company's records show the following selected accounts and amounts for the month...
-
In Buffett s Idaho speech, he said that the advent of the auto meant the death of horses as a form of transport. Can you come up with 1 example or a change you see today and how that could mean...

Study smarter with the SolutionInn App


