Predict the standard potential of each of the following galvanic cells: 3+ (a) Pt(s) Cr+ (aq), Cr+
Question:
Predict the standard potential of each of the following galvanic cells: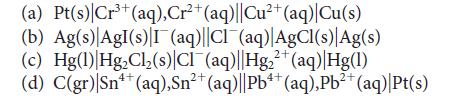
Transcribed Image Text:
3+ (a) Pt(s) Cr³+ (aq), Cr²+ (aq)||Cu²+ (aq) |Cu(s) (b) Ag(s) AgI(s) I (aq)||CI (aq)|AgCl(s) Ag(s) (c) Hg(1) Hg₂Cl₂ (s) CIT(aq) |Hg₂+ (aq) Hg(1) 2+ 4+ (d) C(gr) |Sn¹+ (aq),Sn²+ (aq)||Pb4+ (aq), Pb²+ (aq)|Pt(s) 2+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a 075 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Predict the standard potential of each of the following galvanic cells: 3+ (a) Pt(s)| Fe+ (aq), Fe+ (aq)||Ag* (aq) Ag(s) (b) U(s) U+ aq||V+ (aq) V(s) 2+ (c) Sn(s) Sn+ (aq)||Sn4+ (aq),Sn+ (aq)|Pt(s)...
-
A galvanic cell has the following cell reaction: M(s) + 2 Zn 2+ (aq) 2 Zn(s) + M 4+ (aq). The standard potential of the cell is 10.16 V. What is the standard potential of the M 4+ /M redox couple?
-
Sketch the galvanic cells based on the following half-reactions. Calculate Ïo, show the direction of electron flow and the direction of ion migration through the salt bridge, identify the...
-
Solve: y(4) +18y"+81y = 0 y(0) = -4, y'(0) = 8, y'(0) = 42, y"(0) = -108 - Submit Question X
-
Weighted-average method, spoilage. Appleton Company makes wooden toys in its Forming Department, and it uses the weighted-average method of process costing. All direct materials are added at the...
-
A British hospital chain wishes to make its first entry into the U.S. market by building a medical facility in the Midwest, a region with which its director, Doug Moodie, is comfortable because he...
-
Comment on the phrase information system is an open system.
-
You are provided with the following information for Perkins Inc. for the month ended October 31, 2014. Perkins uses a periodic method for inventory. Instructions (a) Calculate (i) ending inventory ,...
-
What are the interesting objects you have learned about analyzing financial data? Which ratio would you relyon as an investor? as a business manager?
-
Carbonate and hydrogen carbonate (bicarbonate) ions contribute to buffering in a variety of natural systems. You are investigating their role in groundwater percolating through limestone hills into a...
-
Estimate the enthalpy of deprotonation of formic acid at 25C, given that K a = 1.765 * 10 4 at 20C and 1.768 * 10 4 at 30C.
-
Orange Corporation purchased equipment for $30,000. Orange recorded total depreciation of $24,000 on the equipment. On January 1, 2016, Orange traded in the equipment for new equipment, paying...
-
Procedure 4. Find five circular objects with different diameters. These could be objects such as a food or drink cans, balls of different kinds, a broom stick, or any other object around you. A paper...
-
3- 4- Graphing Mass and Volume C.1 Pennies or Marbles Initial volume of water (ml.) Initial mass of cylinder + water (g) C.2 C3 Mass (Pennies or Marbles) 188.3 191.9 g g -1952 199.0 20788 Density g...
-
(k.k) 1 = { $2(09) + bx2({0}) [(*) + 2) = {}]} [ k 3 Fg,2(k1, k2; n) = b1 (n) F2 (k1, k2) + b2(n) +bx(n) (12.87)
-
10A-Lab Report: Simple Harmonic Motion - Mass on a Spring Name: Data Sketch your graph of Position versus Time for Run #1: Enn Data Table 2 Time 3 r Item Run #1 Suspended Mass 0.05 kg Mass of the...
-
1. If the nystatin oral suspension pictured above is used as your source of nystatin for compounding the prescription, how many milliliters of the suspension will you need? Answer: 2. Based on the...
-
McMaster Fabrication has two manufacturing departments: machine operations and assembly. McMaster projected the following numbers for the most recent year. McMaster applies machine operations...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
The following data were observed in an experiment on the photoelectric effect from potassium: Graphically evaluate these data to obtain values for the work function and Plancks constant. 1019 Kinetic...
-
The power (energy per unit time) radiated by a blackbody per unit area of surface expressed in units of W m 2 is given by P = T 4 with = 5.67 10 8 W m 2 K 4 . The radius of the sun is 7.00 10 5...
-
The work function of palladium is 5.22 eV. What is the minimum frequency of light required to observe the photoelectric effect on Pd? If light with a 200. nm wavelength is absorbed by the surface,...
-
Firm E must choose between two business opportunities. Opportunity 1 will generate an $8,000 deductible loss in year 0, $5,000 taxable income in year 1, and $20,000 taxable income in year 2....
-
When discussing the costs of quality, what type of costs are for repairing defects found prior to product delivery? Explain.
-
Built-Tite uses job order costing. The T-account below summarizes Factory overhead activity for the current year. Factory Overhead Debit Credit 17,600 112,200 26,600 61,600 1. Compute total applied...

Study smarter with the SolutionInn App


