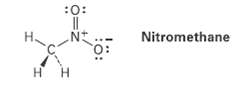
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane
Question:
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO.
Transcribed Image Text:
:0: Nitromethane :O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
Use the formula in Section 23 to confirm the form...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
1. Explain why it must be true, even for someone trying to lose weight, that the last bite of food consumed must have positive marginal utility at a consumer optimum? 2. What is true of the marginal...
-
The compound has a formal charge of (a) -1 on N (b) +2 on N (c) -1 on Al (d) +1 on Br (e) None of the above Br CH3 Br-Al-N-CH.CIH, Br CH3
-
Explain why electric field lines begin on positive charges and end on negative charges. What is the direction of the electric field near positive and negative charges?
-
You are the manager of a large car dealership. At present, you rely on a large pool of dealers to sell your product, but you have noticed recently that consumer opinion appears to be turning away...
-
Roger inherited 100 shares of Periwinkle stock when his mother, Emily, died. Emily had acquired the stock for a total of $60,000 on November 15, 2012. She died on August 10, 2016, and the shares were...
-
Assume that x is a random variable having a Poisson probability distribution with a mean of 1.5. Use statistical software to find the following probabilities: a. P(x 3) b. P(x 3) c. P(x = 3) d. P(x...
-
a. What responsibility does the auditor have for the post audit discovery of facts existing at the date of his report? b. Identify the steps the auditor should take when the client refuses to make...
-
Lisa Vozniak started her own business, We Do Windows. She offers interior and exterior window cleaning for local area residents. Lisa rents a garage to store her tools and cleaning supplies and has a...
-
what is the difference between Activity Based Costing and Traditional Costig ?
-
Jenny Brown opened a law office, on July 1, 2010. On July 31, the balance sheet showed Cash $5,000, Accounts Receivable $1,500, Supplies $500, Office Equipment $6,000, Accounts Payable $4,200, and...
-
Make three-dimensional drawings of the following molecules, and predict whether each has a dipole moment. If you expect a dipole moment, show its direction. (a) H2C = CH2 (b) CHC13 (c) CH2C12 (d) H2C...
-
Calculate formal charges for the non-hydrogen atoms in the following molecules: (a) Diazomethane, (b) Acetonitrile oxide, (c) Methylisocyanide, H2C=N=N: part a H3C-NEC: part b H3C-CEN-o: part c
-
On January 1, 2022, Bryce Inc. changed from the LIFO method of inventory pricing to the FIFO method. Explain how this change in accounting principle should be treated in the companys financial...
-
a. Given that silicon has Young's modulus of about 110 GPa and a density of 2.3 g cm3, calculate the mean free path of phonons in Si at room temperature
-
You are hiking to the top of Mt. Mitchell, NC (elevation 2 km). You start the hike at 0.5 km elevation where the air temperature is 20 C. There are cumulus clouds overhead with the cloud base at 1.5...
-
A bat s echolocation is measured around 9 9 kHz , with a pulse of only 1 2 s . Due to the sampling error, determine a range of possible frequencies.
-
An uncharged 6.6-F capacitor is in series, through a switch, with a 4.10-MQ resistor and a 11.6-V battery (with negligible internal resistance.) The switch is closed at t = 0 and a current of /;...
-
1. A point on the third nodal line from the center of an interference pattern is 35 cm from one source and 42 cm from the second source. The sources are 11.2 cm apart and vibrate in phase at 10.5 Hz....
-
Based on Figure 19, at about what time was the largest number of birds flying over the east-west line marked by the seven cities? Over what part of New York did most of the birds fly? Approximately...
-
What are the four types of poultry production systems? Explain each type.
-
The vapor pressure of some materials can be represented by the equation Values of the constants in this equation are given below. Compute the heats of sublimation of the solids and the heats of...
-
There are two isomeric conjugate acids that produce the following base. Show the structure of each and explain how they produce the same base upon loss of a proton. :0: CH3-C=CH
-
When a is reacted with hydroxide ion, isomer b is formed. (a) Explain why b is favored over a at equilibrium. (b) Show the structure of the conjugate base of a and explain how this isomerization...
-
Compound c is a slightly stronger acid than compound d. The CH3O group has both an inductive effect and a resonance effect on the acidity of d. (a) Explain how the inductive effect of the CH3O group...
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
Evaluate the use of novel catalytic systems, such as structured catalysts, catalytic distillation, and multifunctional catalysts, in achieving process intensification, discussing the effects on...
-
Find the minimum tractive effort required for vehicle to maintain 70mph speed at 5%upgrade through an air density of 0.002045 slug/ft^3. Show all steps and unit conversion please Problem 2:...

Study smarter with the SolutionInn App


