Calculate formal charges for the non-hydrogen atoms in the following molecules: (a) Diazomethane, (b) Acetonitrile oxide, (c)
Question:
Calculate formal charges for the non-hydrogen atoms in the following molecules:
(a) Diazomethane,
(b) Acetonitrile oxide,
(c) Methylisocyanide,
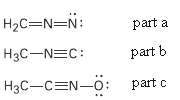
Transcribed Image Text:
H2C=N=N: part a H3C-NEC: part b H3C-CEN-o: part c
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Strategy To find the formal charge of an atom in a molecule follow these two steps 1 Draw an e...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
A few hydrogen and oxygen molecules are introduced into a container in the quantities depicted in the following drawing. The gases are then ignited by a spark, causing them to react and form H2O. a....
-
In the following four models, C atoms are black, H atoms are light blue, O atoms are red, and N atoms are dark blue: a. Write the molecular formula of each molecule. b. Write the condensed structural...
-
True Or False Death benefits are used to compensate the deceaseds family for pain and suffering.
-
Near the end of 2016, Byron realizes that he has a net short-term capital loss of $13,000 for the year. Byron has taxable income (not including the loss) of $123,000 and is single. He owns numerous...
-
Given that x is a hyper geometric random variable, compute p(x) for each of the following cases: a. N = 5, n = 3, r = 3, x = 1 b. N = 9, n = 5, r = 3, x = 3 c. N = 4, n = 2, r = 2, x = 2 d. N = 4, n...
-
What is meant by dual dating an audit report?
-
In early July 2012, Dr. Elaine Matthews separated from her husband of some years. She maintained full custody of the couple's only child, a seven-year-old girl. Since May 1, 1998, Dr. Matthews had...
-
A machine costing $206,800 with a four-year life and an estimated $16,000 salvage value is installed in Luther Company's factory on January 1. The factory manager estimates the machine will produce...
-
On June 30, 2019, Hayes Jewelers purchased inventory of $5,800 on account from Slater Diamonds, a jewelry importer. Terms were 3/15, net 45. The same day Hayes paid freight charges of $400. Upon...
-
Nitromethane has the structure indicated. Explain why it must have formal charges on N andO. :0: Nitromethane :O:
-
Organic phosphate groups occur commonly in biological molecules. Calculate formal charges on the four O atoms in the methyl phosphatedianion. 2- :0: H-C-0-P-0: Methyl phosphate :0:
-
For the Coca-Cola bonds described in BE 124, prepare journal entries to record (a) any unrealized gains or losses occurring in 2024 and (b) the sale of the bonds in 2025, including recognition of any...
-
Identify two digital marketing methods that can be used for brand awareness and support with reasons. Define short-term and long-term goals for brand awareness in the digital marketing strategy. How...
-
How much pressure is applied to the ground by a 56 kg man who is standing on square stilts that measure 0.06 m on each edge? Answer in units of Pa. 003 (part 2 of 2) 10.0 points What is this pressure...
-
Explain how marketing to different countries or cultures may affect your advertising message or campaign? Explain the principle and give examples of actual advertising campaigns localized for...
-
7. A rocket of a mass 200 kg loaded with a propellant of a mass 1200 kg is powered by a rocket engine whose exhaust velocity is 5000 kmph. What is the maximal fuel consumption per second possible,...
-
8. An airplane is flying N75E with an airspeed of 550 mph and a 45 mph wind blowing S75E. What is the actual speed, in mph and direction, written as a bearing, of the plane? Round to the nearest 1...
-
A statistics class consists of 24 students, all but one of whom are unemployed or are employed in low-paying part-time jobs. One student works as an executive secretary earning \($75,000\) per year....
-
What are the three kinds of research types? Explain each type.
-
a. Quantitatively explain why ice skates slide along the surface of ice. Can it get too cold to ice skate? b. Is it possible to ice skate on other materials, such as frozen carbon dioxide? c. What is...
-
Explain why the pKas of compounds near the middle of Table 4.2 are often listed with two figures to the right of the decimal place (that is, for NH4+ the pKa = 9.24), whereas those at the beginning...
-
For each pair of compounds, explain which is the stronger acid?
-
Explain why the compound on the left is a stronger acid than the compound on the right.
-
2-Using the CPT data in figure below, a unit weight of 115 pcf, and an overconsolidation ratio of 3, determine (a) soil behavior type (b) o' using Kulhawy and Mayne correlation and (c) N60 for the...
-
How does the application of computational fluid dynamics (CFD) coupled with multiphase flow modeling enhance the understanding and prediction of complex hydrodynamic behaviors in industrial-scale...
-
1. From the joint distribution defined by the graphical model, determine if the conditional independence holds: ALL FIE, B, and provide brief explanation (5 points). 2. Given an undirected chain...

Study smarter with the SolutionInn App


