Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic
Question:
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich?
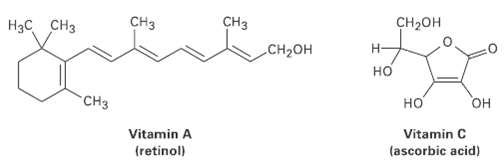
Transcribed Image Text:
CHз CH-он CHз CH2OH H3C CH3 H- но CHз но он Vitamin A (retinol) Vitamin C (ascorbic acid)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
H3C CH3 CH3 CH3 CH3 only one OH group CHOH H CHOH HO several OH ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A soluble salt, MX2, is added to water in a beaker. The equation for the dissolving of the salt is: MX2(s) M2+(aq) + 2X(aq); H > 0
-
The article Modeling Sediment and Water Column Interactions for Hydrophobic Pollutants (Water Research [1984]: 1169 1174) suggests the uniform distribution on the interval from 7.5 to 20 as a model...
-
Water is heated while flowing through a pipe by steam condensing on the outside of the pipe.(a) Assuming a uniform overall heat transfer coefficient along the pipe, derive an expression for the water...
-
For the attractive-nuisance doctrine to apply, the possessor need not be aware that children are likely to trespass on his land or have reason to know that the condition poses an unreasonable risk of...
-
Mandy and Timothy exchange equipment in a like-kind exchange. Mandy receives equipment with a fair market value of $28,000 and transfers equipment worth $20,000 (adjusted basis of $14,000) and cash...
-
According to the Environmental Protection Agency (EPA) Fuel Economy Guide, the 2011 Honda Civic automobile obtains a mean of 36 miles per gallon (mpg) on the highway. Suppose Honda claims that the...
-
What specific procedures should be performed to ascertain the existence of subsequent events?
-
How do the process strategies of eBay and McDonalds differ, and how do their choices relate to customer-introduced variability?
-
The virtual help center for digital scanning equipment produced by High-Tech Company processes 1.2 million customer inquiries per year. Roughly 50% of the time, the chatbot can successfully resolve...
-
On a given PhiladelphiaLos Angeles flight, there are 200 seats. Suppose the ticket price is $475 on average and the number of passengers who reserve a seat but do not show up for departure is...
-
Imidazole forms part of the structure of the amino acid histidine and can acts as both an acid and a base (a) Look at the electrostatic potential map of Imidazole, and identify the most acidic...
-
Fill in the multiple bonds in the following model of naphthalene, C10H8 (gray = C, ivory =H). How many resonance structures does naphthalenehave?
-
Listed below are some common terms of sale. Can you explain what each means? a. 2/30, net 60. b. 2/5, EOM, net 30. c. COD.
-
An explanation for why women remain underrepresented in some areas of employment and are paid less than male counterparts doing the same work is known as what?
-
how do nuanced leadership styles manifest, and what catalytic elements differentiate transformational leadership from transactional leadership in fostering organizational resonance ?
-
Most experts expect the development of worldwide sporting leagues to continue as the cost of communication and transportation continues to fall. Traditional American sports may find themselves in...
-
A single-price monopolist has the following equations representing its marginal cost and demand curves: Q-800-1/3 P MC-20 What are this monopolist's profit maximizing price and quantity?
-
Suppose that you are instead interested in individual-level exposures. Describe a hypothetical individual-level study for a closely related research question. What is your question of interest? How...
-
When a honeybee flies through the air, it develops a charge of +17 pC. How many electrons did it lose in the process of acquiring this charge?
-
A 2500-lbm car moving at 15 mi/h is accelerated at a constant rate of 15 ft/s 2 up to a speed of 50 mi/h. Calculate force and total time required?
-
Prove that C P C V for any fluid, and identify those conditions for which C P = C V .
-
Draw structures for these compounds: (a) 1, 5-Dibromo-2, 2-dichloro-4-ethyl-4-methyl-3-hexanol (b) 2, 2, 5, 6-Tetramethylcyclohex-3-enol (c) tert-Butylamine (d) 1, 2-Cyclopentanediol (e) Dibutyl...
-
Menthol is a component of oil of peppermint. Label each carbon of menthol as primary, secondary, tertiary, or quaternary. Should menthol be classified as a primary, secondary, or tertiary alcohol?...
-
Provide systematic name for these naturally occurring compounds: a) b) c) OH OH OH LL CH-CH-CH OH Isoprene OH Glycerol (obtained from fat) Terpinen-4-ol (a terpene) OH Menthol (a component of...
-
Describe the phenomenon in current accounting practices that has been dubbed the Earnings Game in the article by Justin Fox. Why do companies play this game? Based off this article:...
-
On January 1, 2018, Bark Manufacturing Company Ltd. purchased a machine for $27,500, and expects to use the machine a total of 32,000 hours over the next four years. Bark set the residual value on...
-
Covid and post Covid Economic Recovery for each of the papers you are required to state the following : 1. what was the study about 2. what were the findings 3. list the research gaps and suggest new...

Study smarter with the SolutionInn App


