Imidazole forms part of the structure of the amino acid histidine and can acts as both an
Question:
Imidazole forms part of the structure of the amino acid histidine and can acts as both an acid and a base
(a) Look at the electrostatic potential map of Imidazole, and identify the most acidic hydrogen atom and the most basic nitrogen atom.
(b) Draw structures for the resonance forms of the products that result when Imidazole is protonated by an acid and deprotonated by abase.
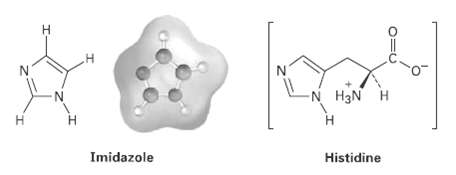
Transcribed Image Text:
N. -N H3N H H. Imidazole Histidine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a The nitrogen on the left is more electronrich ...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The Kitchen Aid brand forms part of the Whirlpool Corporation who are an American manufacturer of appliances based in Michigan in the United States. KitchenAid Australia are a wholesale subsidiary to...
-
Profit serves as both an incentive and a signal. Explain.
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Suppose we have an automatic module on the module path named lizard-^-cricket-^-1.0.0-SNAPSHOT.jar and no Automatic-Module-Name specified. What module name should named modules use to reference it?...
-
Distinguish between a loss that is not recognized on a nontaxable exchange and a loss that is not recognized on the sale or exchange of a personal use asset.
-
The average size CE3 of single-family homes built in the United States is 2,390 square feet (Statistical Abstract of the United States, 2011). A random sample of 100 new homes sold in California...
-
What information should the auditor obtain about litigation, claims, and assessments (LCA)?
-
Dalton Company reports the following operating results for the month of August: Sales $300,000 (units 5,000); variable costs $210,000; and fixed costs $70,000. Management is considering the following...
-
Stacey had $65,286 of income from wages and $298 of taxable interest. Stacey also made contributions of $3,700 to a tax-deferred retirement account. Stacey has 1 dependent and files as married filing...
-
The Trolley Toy Company manufactures toy building block sets for children. Trolley is planning for 2019 by developing a master budget by quarters. Trolley's balance sheet for December 31, 2018,...
-
Using curved arrows, show how the species in part (a) can acts as Lewis bases in their reactions with HC1, and show how the species in part (b) can acts as Lewis acids in their reaction with OH. (a)...
-
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich? CH CH- CH CH2OH H3C CH3 H- CH Vitamin A (retinol) Vitamin C...
-
Figure P3-81 shows a circuit with two sources, a fixed load and a resistor \(R\). Select \(R\) for minimum power transfer to the load. Simulate in Multisim using the "Parameter sweep" under...
-
how do transactional leadership modalities harmonize with transformational leadership imperatives, cultivating a symbiotic relationship between incentivized performance and aspirational visioning in...
-
Write down the dynamic programming recursion that characterizes the optimal behavior of an unemployed worker. Be specific about he assumptions you are making in writing this recursion (and justify...
-
the economic development of foreign countries, including the many obstacles those nations face. For today's discussion, free write on the idea of economic development in foreign nations. Why have...
-
what synergistic interplay emerges between laissez-faire leadership proclivities and participative leadership tenets, fostering an ecosystem of innovation and adaptive resilience ? Explain
-
Would a 5% or 10% increase in the price of a product lead you to switch to another product? For what fraction of purchases? Would an increase in the price of a broader collection of products (such as...
-
Researchers doing statistical studies in biology, psychology, and sociology are grateful for the existence of twins. Twins can be used to study whether certain traits are inherited from parents...
-
Integration is a vital concept when applied in one?s life. Integrating your life means making ideal choices. Perfect choices on the other go in line with quality decisions. Quality decisions lead to...
-
The triple point of iodine, I 2 , occurs at 112.9C and 11.57 kPa. The heat of fusion at the triple point is 15.27 kJ/mol, and the following vapor pressure data are available for solid iodine:...
-
Draw compounds that meet these requirements: (a) A primary alcohol (b) A tertiary alcohol (c) A secondary alkyl chloride (d) A secondary amine
-
What is wrong with the name given for these compounds? Provide the correct name for each. a) c) 18 2-Ethyl-2-pentene CHCHCH 3-Cyclohexylpropane 1-Chloro-2-cyclopentene OH 2-2-Methylbutylcyclopentanol...
-
Name these compounds: a) ) g) 1) k) - OH NH b) CI d) f) CH=CHCHC=CH h) Br j) O OCH 3
-
Mrs. Sam Taffer was a brilliant engineer at IBM and made a lot of money from stock options. Since then, she has worked as a musician and raised her two sons. She wishes to have most of her property...
-
Miller Company ended its fiscal year on June 30, 2017. The company's adjusted trial balance as of the end of its fiscal year is shown below. MILLER COMPANY Adjusted Trial Balance June 30, 2017...
-
What depreciation method is used to write off property, plant and equipment? Regarding the triple bottom line, what information did the company disclose about environmental matters in the reports?...

Study smarter with the SolutionInn App


