On reaction with Cl 2 in the presence of light, an unknown compound with the formula C
Question:
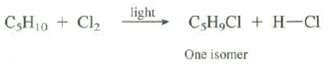
On reaction with Cl2 in the presence of light, an unknown compound with the formula C5H10 gives only one isomer of C5H9Cl (see problem 2.39). What is the DU of the unknown compound? Show the structure of the unknown compound and the product of its reaction with Cl2.
Transcribed Image Text:
CsH₁0 + Cl₂ light C,H,Cl + H-CI One isomer
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The DU of C5H0 25 2 122 1 so the unknown has one rin...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
An unknown compound of molecular formula C5H9NO gives the IR and NMR spectra shown here. The broad NMR peak at δ7.55 disappears when the sample is shaken with D2O. Propose a structure,...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or...
-
Marcus is the HR manager for United Airlines, an Illinois-based company. One of his employees has recently become disabled and is unable to fulfill the essential functions of his current position,...
-
What are the advantages and disadvantages of basing individual incentives on company-wide performance?
-
The Excel file Salary Data provides information on current salary, beginning salary, previous experience (in months) when hired, and total years of education for a sample of 100 employees in a firm....
-
Uniform flow in a sluggish channel having a nearly rectangular cross section that is \(498 \mathrm{ft}\) wide and \(16.5 \mathrm{ft}\) deep carries a flow of \(8,250 \mathrm{ft}^{3} / \mathrm{s}\)....
-
Haldane Company estimates that its overhead costs for 2014 will be $360,000 and output in units of product will be 300,000 units. Required a. Calculate Haldanes predetermined overhead rate based on...
-
A tensile load is applied to a thin-walled hollow cylinder. Determine the change in wall thickness and in mean radius at the point of maximum load, if the stress curve is given by = Ae", where is the...
-
In a certain developing country, ecotourism income has been increasing in recent years. The income y (in thousands of dollars) x years after 2000 can be modeled by y = 1.15x + 14. (a) Draw the graph...
-
One of the isomers of C 5 H 12 reacts with Cl 2 in the presence of light to produce three isomers of C 5 H 11 Cl: This reaction replaces am one of the hydrogen?s of C 5 H 12 with a Cl. What arc the...
-
Explain how the dipole moment for CH3Cl ( = 1.9 D) can be larger than the dipole moment for CH3F ( = 1.8 D).
-
Firm A plans to acquire Firm B. The acquisition would result in incremental cash flows for Firm A of $10 million in each of the first five years. Firm A expects to divest Firm B at the end of the...
-
Give some examples of requirements where they define both objectives and constraints.
-
When is the best time to gather requirements about a product?
-
How do you mine use cases to find classes?
-
Looking at the chart of vaccine-preventable contagious diseases, provide a prioritized list of required vaccinations for health care employees. Be sure to provide a rationale for your rankings, as...
-
A sport organization has a commitment from a sponsor for a $17,000 payment in three years. What is the present value of that money if it is discounted at (a) 3%, (b) 5%, and (c) 9%?
-
Refracted light that bends away from the normal is light that has (a) slowed down. (b) speeded up. (c) bounced. (d) diffracted.
-
Draw two scatterplots, one for which r = 1 and a second for which r = 21.
-
A force of 40 N is required to hold a spring that has been stretched from its natural length of 10 cm to a length of 15 cm. How much work is done in stretching the spring from 15 cm to 18 cm?
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
P-Nitro benzyl alcohol is more acidic than benzyl alcohol but p-methoxy benzyl alcohol is less acidic. Explain.
-
Predict the products of the following reactions: CH (a) 1. CHCH2- 2. NaOH, H2O2 "CH (b) 1. HglOAc)2. 0 2. NABH4 (c) CCH2CH2CH2 CH2CH2CH2CH3 C=C 1. Os04 2. NaHSO3, H20 -
-
Using the following information: a. The bank statement balance is $3,048. b. The cash account balance is $3,300. c. Outstanding checks amount to $755. d. Deposits in transit are $809. e. The bank...
-
Determine the average tax rate and the marginal tax rate for each of the following instances: Use the Tax Tables for taxpayers with taxable income under $ 1 0 0 , 0 0 0 and the Tax Rate Schedules for...
-
A machine was bought on 1 st January 2 0 1 7 for $ 1 2 , 0 0 0 . The policy is to depreciate the machine at 1 0 % on reducing balance method at the end of each year, ending on 3 1 st December....

Study smarter with the SolutionInn App


