Predict the products of the following reactions: CH (a) 1. CHCH2- 2. NaOH, H2O2 CH (b)
Question:
Predict the products of the following reactions:
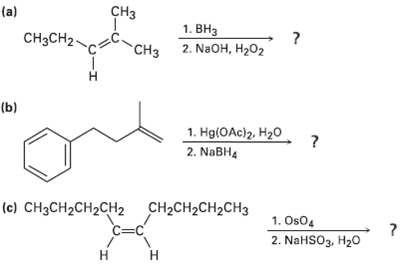
Transcribed Image Text:
CHз (a) 1. ВНз CHзCH2- 2. NaOH, H2O2 "CHз (b) 1. HglOAc)2. Нг0 2. NABH4 (c) CНзCH2CH2CH2 CH2CH2CH2CH3 C=C 1. Os04 2. NaHSO3, H20 Н н -т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a CH3 CH3CHCHCHCH3 CH3 OH 2Methyl3pentanol Remember that the hydroxyl group is bonded t...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the products of the following reactions (the aromatic ring is un-reactive in all cases). Indicate region-chemistry when relevant. H2/Pd (a) Br2 (b) Os04 (c) NMO Cl2, H20 (d) CH212, Zn/Cu (e)...
-
Predict the products of the following reactions: (a) (b) CH CH3CH2CH-0-CH2CH2CH3 CH r HBr
-
Predict the products of the following reactions: CH (b) CH (a) 1. (NH2)2C=S 2. NaOH, H20 -CH CH2CH2CH2Br Hr SCH2CH3 (d) (c) Br2, ? H0z. 2 SH
-
Ava bought a house on 1 July 2000 for 62,000. She occupied the entire property as her PPR until 1 August 2007 when she began using one-quarter of the house for business purposes. This continued until...
-
Explain each traditional organizational structure.
-
Show that if f : A B and G, H are subsets of B, then f-1(G H) = f-1 (G) f-1(H) and f-1 (G H ) = f-1 (G) f-1(H).
-
Anthony, CPA, prepared the flowchart on page 349 which portrays the raw materials purchasing function of one of Anthony's clients, a medium-sized manufacturing company, from the preparation of...
-
Davis Fabricators buys metal for manufacturing from two suppliers, Alpha Metals and First Parts. If the metal is delivered late, the shipment to the customer is delayed. Delayed shipments lead to...
-
The potential of the two-barrier system is given by U(z) = 8(z+a) + a8(z - a). 1. Find the wavefunction of the system at energy E. 2. The amplitude of the wavefunction at z>a is the transmission...
-
Built-Tight is preparing its master budget for the quarter ended September 30. Budgeted sales and cash payments for product costs for the quarter follow. Sales are 20% cash and 80% on credit. All...
-
P-Nitro benzyl alcohol is more acidic than benzyl alcohol but p-methoxy benzyl alcohol is less acidic. Explain.
-
What reagent would you use to accomplish each of the following reactions? , 2. (a) CHCH-CH2co CHH2CH2cH , 2. (b) CHH2H2H2 CHH2H2cH (c)
-
What are the four bases for determining employee gross earnings?
-
According to Maslows theory of needs, what is the order in which individuals satisfy their needs?
-
Define and thoroughly explain what the Trade Dispute Act is and its purpose.
-
What is Media? And describe how Media can adding value and sentiment to society.
-
What are the structural and functional properties of G protein-coupled receptors (GPCRs) that enable their diverse signaling capabilities, and how do these receptors serve as pivotal mediators of...
-
How does the intricate interplay between chromatin remodeling complexes and epigenetic modifications regulate gene expression in multicellular organisms, and what implications does this have for...
-
Je Ju Consultants had the following adjusted trial balance (amounts in thousands). Instructions a. Prepare closing entries at June 30, 2025. b. Prepare a post-closing trial balance. Je Ju Consultants...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
It is sometimes necessary to produce saturated steam from superheated steam (steam at a temperature higher than the vapor-liquid coexistence temperature at the given pressure). This change can be...
-
Rewrite each of the following reactions using curved arrows and show all nonbonding electron pairs: (a) (b) (c) CH NH2 HC
-
Follow the curved arrows and write the products. (a) (b) (c) (d) + BF3 BFs
-
Write an equation, using the curved-arrow notation, for the acid-base reaction that will take place when each of the following are mixed. If no appreciable acid-base reaction takes place, because the...
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


