One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2.
Question:
One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (1) Protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+.
(a) Draw two resonance structures of diazomethane, and account for step 1.
(b) What kind of reaction occurs in step2?
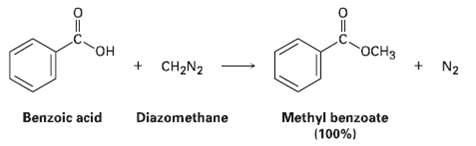
Transcribed Image Text:
OCH3 + N2 он + CH2N2 Methyl benzoate (100%) Benzoic acid Diazomethane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a HCNEN HCNN Resonance forms show that the carbon of diazo...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Diazomethane can be used to convert a carboxylic acid into a methyl ester. Propose a mechanism for this reaction. a carboxsyllc diazomethane RCOH + CH2N2 RCOCH3 N2 a methyl acid ester
-
Draw two resonance structures for diazomethane, CH2N2. Show formal charges. The skeletal structure of the molecule is C N N
-
A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons. (a) Draw the resonance forms of a carboxylic acid that is protonated on the hydroxyl oxygen atom. (b) Compare the...
-
The University of Professional Studies, Accra (UPSA) is a public university in Ghana. UPSA is the first university in Ghana to provide both academic and business professional education. The...
-
Are you a member of the "thumb generation"? Can you work the keyboard of your smartphone faster than most people can speak? The term thumb generation was coined in South Korea and Japan and is...
-
For waiting time 2. Use the method of maximum likelihood to estimate the rate λ from the accompanying table of data drawn from an exponential distribution. In each case, find the...
-
Explain the nature of the first six types of Braitenberg vehicles. Discuss how these vehicles can help us to understand the nature of intelligence.
-
Stuckey, Inc., manufactures industrial 55 gallon drums for storing chemicals used in the mining industry. The body of the drums is made from aluminum and the lid is made of chemical resistant...
-
Use the simplex method to maximize the function under the given constraints. (If the solution is unbounded, enter INFINITY.) Maximize f = 3x + y subject to 4x + y 44 4x + 3y 60 x 0, y 0. f = INFINITY
-
a. Draw a UML class diagram that describes the Tablet Store's purchases and payments process. b. Using Microsoft Access, implement a relational database from your UML class diagram. Identify at least...
-
Tranexamic acid, a drug useful against blood clotting, Is prepared commercially from p-methyl Benzonitrile. Formulate the steps likely to be used in the synthesis. (Don?t worry about cis-trans...
-
The hydrolysis of a biological thioester to the corresponding carboxylate is often more complex than the overall result might suggest. The conversion of succinyl CoA to succinate in the citric add...
-
For each of the following capital asset dispositions, determine whether the taxpayer has realized a gain or loss on the disposition and whether that gain or loss is short-term or long-term. a. Ari...
-
Compare Pfizers 2 0 2 2 and 2 0 2 3 balance sheet and income statement. Write a comment and comparison for each of the financial comparison Examine economic conditions and financial conditions in 2 0...
-
Part II-Computing Starbucks' Share Value Using Free Cash Flows to All Debt and Equity Stakeholders g. At the end of 2012, Starbucks had $1,263 million in outstanding interest-bearing short-term and...
-
How is share price of a stock - exchange - listed corporation estimated using the earnings multiple?
-
A cost control report of a certain contract gives the following figures as percentages of the contract a completion budgeted cost: Budgeted Cost of Work Scheduled = 150,000 L.E - Actual Cost of Work...
-
Assess how the 1 3 8 Student Living Jamaica ltd has changed from 2 0 2 1 - 2 0 2 2 . What conclusions can you draw from this information about 1 3 8 Student Living Inc. strategy? 5 . What conclusions...
-
The chemical formula for quartz is SiO2. What is the chemical formula of coesite, a polymorph of quartz?
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Write a short paragraph describing chemical bonding according to the Lewis model, valence bond theory, and MO theory. Indicate how the theories differ in their description of a chemical bond and...
-
Explain each of the following observations. The allene 2, 3-heptadiene can be resolved into enantionmers, but the cumulene 2, 3, 4-heptatriene cannot.
-
Using the Huckel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the -electron count in each case. (a) (b)
-
The following compound is not aromatic even though it has 4n + 2 electrons in a continuous cyclic array. Explain why this compound is not aromatic.
-
4 The manager of the Fleet division of Potlatch Automotive is evaluated on her division's return on investment and residual income. The company requires that all divisions generate a minimum return...
-
At the end of January of the current year, the records of Donner Company showed the following for a particular item that sold at $16 per unit: Transactions Units Amount Inventory, January 1 Purchase,...
-
During Year 1, Chung Corporation earned $6,900 of cash revenue and accrued $3,800 of salaries expense. Required: (Hint. Record the events in general ledger accounts under an accounting equation...

Study smarter with the SolutionInn App


