Tranexamic acid, a drug useful against blood clotting, Is prepared commercially from p-methyl Benzonitrile. Formulate the steps
Question:
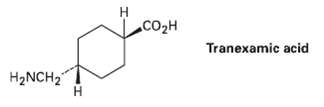
Tranexamic acid, a drug useful against blood clotting, Is prepared commercially from p-methyl Benzonitrile. Formulate the steps likely to be used in the synthesis. (Don?t worry about cis-trans isomers; heating to 300 ?C interconvert?s the isomers.)
Transcribed Image Text:
H. CO2H Tranexamic acid H2NCH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (18 reviews)
OH NBS excess P PhCO22 NH3 CHBr CN CH3 H3O OH CH3 OH HOC H H RhC CHNH H CH2NH2 ...View the full answer

Answered By

Amos Kiprotich
I am a wild researcher and I guarantee you a well written paper that is plagiarism free. I am a good time manager and hence you are assured that your paper will always be delivered a head of time. My services are cheap and the prices include a series of revisions, free referencing and formatting.
4.90+
15+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The choice of the measurement model to be used in Financial Statements has always been particularly complex and controversial within the academic community, standard setters and practice. In a recent...
-
The dielectric to be used in a paral1el-plate capacitor has a dielectric constant of 3.60 and a dielectric strength of 1.60 X 107 V/m. The capacitor is to have a capacitance of 1.25 X 10- 9 F and...
-
The availability and form of data to be used in preparing a forecast are often seen as especially critical areas. Summarize, in your own words, the database considerations in the forecasting process...
-
Tri-State Railroad Company organizes its three divisions, the Southeast (SE), East (E), and South (S) regions, as profit centers. The chief executive officer (CEO) evaluates divisional performance,...
-
Select four graphics from newspapers or magazines, in hard copy or online. Look in The Wall Street Journal, USA Today, BusinessWeek, U.S. News & World Report, Fortune, or other business news...
-
Twenty events occur in 1 min. Compare the likelihood with the maximum likelihood estimator of with the likelihood if = 10.0. Find the likelihood as a function of the Poisson parameter , find the...
-
Think of a real-world interface agent. Discuss to what extent this agent has autonomy, learning abilities, and intelligence.
-
The net income reported on the income statement for the current year was $92,000. Depreciation recorded on store equipment for the year amounted to $18,600. Balances of the current asset and current...
-
The crew of a helicopter need to land temporarily in a forest and spot a flat horizontal piece of ground (a clearing in the forest) as a potential landing site, but are uncertain whether it is wide...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
N, N-Diethyl-m-toluamide (DEFT) is the active ingredient in many insect- repellent preparations. Flow might you synthesize this substance fromm-bromotoluene? . CH2CH3 N,N-Diethyl-m-toluamide CH-CH
-
One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (1) Protonation of diazomethane by the carboxylic...
-
Stock prices: Following are the closing prices of Microsoft stock for each trading day in May and June 2013. a. Find the mean and median price in May. b. Find the mean and median price in June. c....
-
4. The assets listed below belongs to Paradise Ltd and the information for each asset on 31 December 2011 are given. ACCUMULATED DEPRECIATION ASSET COST MOTOR RM 258400 RM 119000 VEHICLE DEPRECIATION...
-
On November 1 st of the current year Elwha Corporation took out a $ 5 0 , 0 0 0 , 3 year loan with an interest rate of 7 % . Elwha Corporation will pay all the interest and principal at the end of...
-
Prepare a tax memo using the information below in the format of Facts, Issue, Conclusion, Authorities, analysis and summary, and actions to be taken. Jim just moved out on his own after graduating...
-
The following condensed income statements of the Jackson Holding Company are presented for the two years ended December 31, 2024 and 2023: Sales revenue Cost of goods sold Gross profit Operating...
-
TDB Limited has prepared the following information for the current year. The company has paid all income taxes related to prior years but has paid no income tax for the current year. Income from...
-
Why should mines be air-conditioned?
-
Write a while loop that uses an explicit iterator to accomplish the same thing as Exercise 7.3. Exercise 7.3. Write a for-each loop that calls the addInterest method on each BankAccount object in a...
-
A molecule with the formula AB 3 has a trigonal pyramidal geometry. How many electron groups are on the central atom (A)?
-
Which d the following species should be aromatic by the Huckel 4n + 2 rule? (a) (b) (c) N: isoxazole C2Hs
-
Use the curved-arrow or fishhook notation to derive the major resonance structures for each of the following species. Determine which, if any, structure is the most important one in each case. (a)...
-
What six-carbon conjugated diene would give the same single product from either 1, 2- or 1, 4-addition of HBr?
-
A description of how each of the two management styles selected distinctively create organizational cultures. Describe the strengths and weaknesses of the two styles. Explain how these differences...
-
A moon orbits a giant planet in a circular orbit with a semi-major axis a = 2.0473 x 10^8 metres and an orbital period P = 2.1299 days. What is the mass of the giant planet in kilograms to five...
-
Using the right-hand rule, in which direction will the magnetic force act on a positively charged particle that is moving to the left and experiencing a magnetic field straight ahead? a. Using the...

Study smarter with the SolutionInn App


