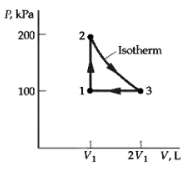
One mole of an ideal monatomic gas at an initial volume V1 = 25 L follows the
Question:
One mole of an ideal monatomic gas at an initial volume V1 = 25 L follows the cycle shown in figure. All the processes are quasi-static. Find
(a) The temperature of each state of the cycle,
(b) The heat flow for each part of the cycle, and
(c) The efficiency of thecycle.
Transcribed Image Text:
P, kP | 200 2. Isotherm 100 2V1 V,L V1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
a Using PV nRT T 1 100 258314 K 3007 K T 2 T 3 6014 K b Q 1...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
Consider one mole of an ideal monatomic gas at 300K and 1 atm. First, let the gas expand isothermally and reversibly to twice the initial volume, second, let this be followed by an isentropic...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
A heat engine follows the cycle shown in the figure. (a) How much net work is done by the engine in one cycle? (b) What is the heat flow into the engine per cycle? 4.00 atm 1.00 atm 0.200 m 0.800 m3 T
-
In Exercises find the indefinite integral. sech(2x1) dx
-
Data concerning a recent period's activity in the Assembly Department, the first processing department in a company that uses the FIFO method in its process costing, appear below: A total of 26,000...
-
For each of the following situations, use the IS-LM-FX model to illustrate the effects of the shock and the policy response. Note: Assume the government responds by using monetary policy to stabilize...
-
Identify each of the following as an example of a perceived pressure (P), perceived opportunity (O), or rationalization (R) in the fraud triangle: 1. Job dissatisfaction 2. Greed. 3. "It's for a good...
-
Presented below are the financial statements of Newman Company. Additional data: 1. Dividends declared and paid were $27,000. 2. During the year equipment was sold for $8,500 cash. This equipment...
-
Critically evaluate the business operations of Starbucks, Make recommendations how can the business operations could be improved
-
On January 1, 2019, Aspen Company acquired 80 percent of Birch Companys voting stock for $288,000. Birch reported a $300,000 book value, and the fair value of the noncontrolling interest was $72,000...
-
An engine using 1 mol of an ideal gas initially at V 1 = 24.6 L and T = 400 K performs a cycle consisting of four steps: (1) an isothermal expansion at T = 400 K to twice its initial volume, (2)...
-
An ideal gas (? = 1.4) follows the cycle shown in figure. The temperature of state 1 is 200 K. Find (a) The temperatures of the other three states of the cycle and (b) The efficiency of the cycle. P,...
-
What are three main characteristics of the capitalist economy that make possible cycles of boom and bust?
-
Pure Fiji state How long have they been operational? Where is this company located (headquarters and all its branches) ? Which SM platforms does this company have their presence on? Provide these SM...
-
Choose a program within a human services agency for this assignment and describe how you might fund one or more of its programs.
-
Describe a health care improvement project in which you were involved (e.g. improvement in patient satisfaction scores). Explain how the project was organized and how clear directions were provided...
-
what is the monthly payment for a car purchased at $24,500 when terms are 4 years loan , 3.99% interest rate? No money down payment. 2.) given the following information calculate the expected return...
-
Discuss your thoughts on collective bargaining and its pros and cons.?
-
What are the various types of chain drive? Explain with neat sketches.
-
The cash records of Holly Company show the following four situations. 1. The June 30 bank reconciliation indicated that deposits in transit total $720. During July, the general ledger account Cash...
-
Under what conditions will a bond sell at a premium above par? At a discount from par?
-
The electric field at a distance of 0.145 m from the surface of a solid insulating sphere with radius 0.355 m is 1750 N/C. (a) Assuming the sphere's charge is uniformly distributed, what is the...
-
A conductor with an inner cavity, like that shown in Fig. 22.23c, carries a total charge of + 5.00 nC. The charge within the cavity, insulated from the conductor, is -6.00 nC. How much charge is on...
-
Apply Gauss's law to the Gaussian surfaces S2, S3, and S4 in Fig. 22.21b to calculate the electric field between and outside the plates.
-
The company has 200 000 non-redeemable preference shares in issue with a par value R100. Preference dividends are payable annually in arrears. The non-redeemable preference shares are currently...
-
The total interest paid was $3,455,854.51. The quarterly installments were $181,861.82. The value of the loan was $2,000,000. The interest rate is not known, but you do know the straightforward...
-
Your company's finance manager has asked you to analyze the advisability of selling a piece of land owned by the company. He has decided that he will only proceed to authorize the sale if the land...

Study smarter with the SolutionInn App


