One of the reactions used in determining the sequence of nucleotides in a strand of DNA is
Question:
One of the reactions used in determining the sequence of nucleotides in a strand of DNA is reaction with hydrazine. Propose a mechanism for the following reaction, which occurs by an initial conjugate addition followed by internal amideformation.
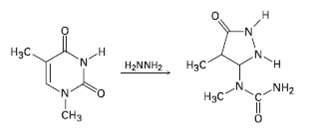
Transcribed Image Text:
Нас. Нас H. H2NNH2 NH2 N. Нас CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
HC NH NH CHO NH H3C HN HN HC H3CN N CH3 NH 1 N...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. Electron flow arrows should start on the...
-
Consider the following hypotheses: H 0 : = 140 H 1 : 140 Given that x = 148.1, s = 37.5, n = 20, and = 0.02, answer the following questions: a. What conclusion should be drawn? b. Use PHStat to...
-
Rewrite this rsum so that it follows the guidelines presented in this chapter. Sylvia Manchester 765 Belle Fleur Blvd. New Orleans, LA 70113 (504) 312-9504 smanchester@rcnmail.com PERSONAL: Single,...
-
How many grams of Na2CO3 (FM 105.99) should be mixed with 5.00 g of NaHCO3 (FM 84.01) to produce 100 mL of buffer with pH 10.00?
-
What three responsibilities do generally accepted auditing standards impose in compliance audits of governmental entities?
-
The bookkeeper for Joes Hardware Co. records all revenue and expense items in nominal accounts during the period. The following balances, among others, are listed on the trial balance at the end of...
-
A particle of dust lands 46.4 mm from the center of a compact disc (CD) that is 120 mm in diameter. The CD speeds up from rest, and the dust particle is ejected when the CD is rotating at 96.0...
-
Suppose the S&P 500 Index portfolio pays a dividend yield of 2% annually. The index currently is 3,000. The T-bill rate is 3%, and the S&P futures price for delivery in one year is $3,045. Construct...
-
?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous...
-
Which compound in each of the following pairs is more basic? (a) CH 3 CH 2 NH 2 or CH 3 CH 2 CONH 2 (b) NaOH or CH 3 NH 2 (c) CH 3 NHCH 3 or pyridine
-
In determining whether or not a variable interest entity is to be consolidated, the FASB focused on a. Legal control. b. Share of profits and obligation to absorb losses. c. Frequency of intercompany...
-
Let : R R ' be a ring homomorphism. Prove that I is an ideal of R then ( I ) is an ideal of ( R ) .
-
Find the root if it is a real number. - 144
-
List all the factor pairs and find the sum of each pair. List all the factor pairs of 18 and find the sum of each pair. Note that because the required sum, -11 , is negative and 18 is positive, both...
-
Find the root if it is a real number. 3 -343
-
What are the two solutions to x + 1 = 0? Write complex number solutions as a + ib.
-
The technetium isotope \({ }^{99} \mathrm{Tc}\) is useful in medical imaging, but its short \(6.0 \mathrm{~h}\) halflife means that shipping it from a source won't work; it must be created where it...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
Which acid has the largest K a : HClO 2 (aq), HBrO 2 (aq), or HIO 2 (aq)? a) HClOz(aq) b) HBrO (aq) c) HIO(aq) d) All three acids have the same Ka.
-
Draw all reasonable electron-dot formulas for the nitronium ion (NO2+), the electrophile in aromatic nitrations. Show any formal charges. Which structure is favored and why? (See Secs. 1.11 and 1.12.)
-
How might Kekule explain the fact that there is only one dibromobenzene with the bromines on adjacent carbon atoms, even though we can draw two different structures, with either a double or a single...
-
Write out all steps in the mechanism for the reaction of a. H,C CH3 +nitric acid (H SO, catalyst) b CH +t-butyl chloride +AlCl
-
Assume an organization needs to do mass layoffs in order to counteract the slowing market demand for their product. What would be considered going above and beyond legal duty to do the right thing by...
-
We are in an incredibly stressful time in our lives. Watch the Ted Talk, "How Burnout Makes Us Less Creative". Share your thoughts on the video. Then discuss what are some actions that you can take...
-
Image transcription text QUESTION 1 Diffusion (a) The diffusion coefficients for carbon in nickel are given at two temperatures as shown in Table 1: Table 1 diffusion coefficients for carbon in...

Study smarter with the SolutionInn App


