?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is
Question:
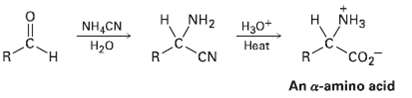
?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous acid. Propose a mechanism for the reaction.
Transcribed Image Text:
н NH2 H NH3 Нао* NH,CN Нао Heat CN co2 An a-amino acid R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
NH NH3 addition of NH3 COH H R NH3 H ...View the full answer

Answered By

Cyrus Sandoval
I a web and systems developer with a vast array of knowledge in many different front end and back end languages, responsive frameworks, databases, and best code practices. My objective is simply to be the best web developer that i can be and to contribute to the technology industry all that i know and i can do. My skills include:
- Front end languages: css, HTML, Javascript, XML
- Frameworks: Angular, Jquery, Bootstrap, Jasmine, Mocha
- Back End Languages: Java, Javascript, PHP,kotlin
- Databases: MySQL, PostegreSQL, Mongo, Cassandra
- Tools: Atom, Aptana, Eclipse, Android Studio, Notepad++, Netbeans.
Having a degree in Computer Science enabled me to deeply learn most of the things regarding programming, and i believe that my understanding of problem solving and complex algorithms are also skills that have and will continue to contribute to my overall success as a developer.
I’ve worked on countless freelance projects and have been involved with a handful of notable startups. Also while freelancing I was involved in doing other IT tasks requiring the use of computers from working with data, content creation and transcription.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following amines can be prepared by the Gabriel synthesis? Which ones cannot? Write equations showing the successful applications of this method. (a) Isobutylamine (b) tert-Butylamine...
-
Propose a mechanism for the reaction of benzoic acid with oxalyl chloride. This mechanism begins like the thionyl chloride reaction, to give a reactive mixed anhydride. Nucleophilic acyl substitution...
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Dr. Thomas Bilski is a primary care physician with a practice that is currently serving 280 patients. Dr. Bilski would like to administer a survey to his patients to measure their satisfaction levels...
-
Using your team's answers to Activity 4, make the statements stronger by quantifying them (make up any numbers you need).
-
Calculate the quotient [HCO-2 ]/[HCO2H] at pH 3.744 if the ionic strength is 0.1 M by using the effective equilibrium constant listed for = 0.1 in 0.1 in Appendix G.
-
What is the auditor's responsibility under generally accepted auditing standards to detect direct and material violations of laws and regulations?
-
Refer to Problem E.5. Beth Zion Hospital has just been informed that only the first 10 transplants must be performed at the hospitals expense. The cost per hour of surgery is estimated to be $5,000....
-
A stellar black hole may form when a massive star dies. The mass of the star collapses down to a single point. Imagine an astronaut orbiting a black hole having five times the mass of the Sun. Assume...
-
Elizabeth Burke wants to develop a model to more effectively plan production for the next year. Currently, PLE has a planned capacity of producing 9,100 mowers each month, which is approximately the...
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
One of the reactions used in determining the sequence of nucleotides in a strand of DNA is reaction with hydrazine. Propose a mechanism for the following reaction, which occurs by an initial...
-
On November 30, the end of the current fiscal year, the following information is available to assist Allerton Companys accountants in making adjusting entries: a. Allertons Supplies account shows a...
-
UQ 6. Identify at least ten different kinds of data files. For each file you identify, indicate whether it would be found in the accounting system for the following organizations: a. service...
-
Question 4 - Determine After-Tax Income During the year Soylent, Inc. (a publicly traded, non-closely held corporation) presents the following information related to its operations: Total Sales...
-
Solve by factoring: 3x - 13x + 4 = 0 List the solutions, separated by commas.
-
Does the exponential expression represent growth or decay? 0.92(1.006)' The given exponential expression represents
-
A garden hose can fill a swimming pool in 9 days, and a larger hose can fill the pool in 4 days. How long will it take to fill the pool if both hoses are used?
-
Many medical PET scans use the isotope \({ }^{18} \mathrm{~F}\), which has a half-life of \(1.8 \mathrm{~h}\). A sample prepared at 10:00 A.M. has an activity of \(20 \mathrm{mCi}\). What is the...
-
Dawson Companys balance sheet information at the end of 2019 and 2020 is as follows: Additional information: The company did not issue any common stock during 2020. Required : Next Level Fill in the...
-
Define pH. What pH range is considered acidic? Basic? Neutral?
-
The structure of the nitro group (-NO2) is usually shown as Experiments show that the two nitrogen-oxygen bonds have the same length of 1.21 Ã. This length is intermediate between 1.36...
-
Draw all reasonable resonance structures for naphthalene (C10H8, Sec. 4.13) and rationalize the different bond lengths for the C-C bonds.
-
When naphthalene is hydrogenated, the heat released is about 80 kcal/mol. Using an isolated cyclohexene unit for comparison, estimate the resonance energy of naphthalene. Why is the resonance energy...
-
In what ways does HR assist managers in handling employee performance issues or conflicts within their teams? What role does HR play in helping managers navigate challenges related to allegations and...
-
Why is A/B testing so useful for marketing, and digital marketing in particular? Give at least two examples of A/B testing for marketing purposes. Give a null and alternative hypothesis for one of...
-
Define the following sets (30 points) (a) X = {(x, y) = R: y = f(x) = 3x + 3y 42} =1 (b) Y={(x, y) = R: y = f(x) = 2x + 2y 42} (c) Z = XUY (d) Prove whether or not each set defined above is convex?

Study smarter with the SolutionInn App


