One of these isomers is miscible with water, and the other is nearly insoluble. Explain. CHCHCHCOH CH,COCHCH3
Question:
One of these isomers is miscible with water, and the other is nearly insoluble. Explain.
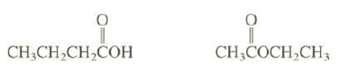
Transcribed Image Text:
CH₂CH₂CH₂COH CH,COCH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
The carboxylic acid is miscible with water be...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
There are two stereoisomers of 1-tert-butyl-4-chlorocyclohexane. One of these isomers reacts with sodium ethoxide in an E2 reaction that is 500 times faster than the reaction of the other isomer....
-
Water and glycerol, CH2(OH)CH(OH)CH2OH, are miscible in all proportions. What does this mean? How do the OH groups of the alcohol molecule contribute to this miscibility?
-
Explain why water forms nearly spherical droplets on the surface of a freshly waxed car. Why doesn't water bead on a clean windshield?
-
An investor has $60,000 to invest in a $280,000 property. He can obtain either a $220,000 loan at 9.5 percent for 20 years or a $180,000 loan at 9 percent for 20 years and a second mortgage for...
-
Assume that you have just been employed at Southern Coast Wealth Foods as an assistant and you are concerned with the high level of reliance on PCI to evaluate and compare the performance of the...
-
Refer to Exercise 17.27. a. Estimate the variance components in this study. b. Proportionally allocate the sources of variability with respect to the total variability in solder strength. c. Place a...
-
At the Diabetes Institute, patients are classified as compliant (they take their medication as they should) or not (they don't). A new program manager has a new idea: Have some patients continuously...
-
Goldstein Inc. manufactures and sells plastic boxes and trays. Sales are projected to be evenly spread over the annual period. Estimated product sales and material needs for each unit of product...
-
You create a caller ID policy named Policy1. You need to configure Policy1 to block outbound caller ID information. What should you do in Policy1? Explaain
-
Your brother has just started a new job as the Controller of an IESBA restricted audit client. You do not serve on the audit engagement. What steps must you take to ensure your independence is not...
-
Benzene and hexane are both liquids at room temperature. Do you expect benzene and hexane to be miscible? Do you expect benzene and water to be miscible? Explain. Hexane 0 Benzene
-
Because of two hydrogen bonds, carboxylic acids show a very strong attractive force between two molecules that persists even in the gas phase. Show this hydrogen bonding between two carboxylic acid...
-
A footwear manufacturing machine manufactures each piece separately. Suppose pairs are manufactured, with the following results obtained for the range (number). Perform an appropriate analysis of...
-
In the past seven years, Sarah's uncle has been paying her monthly allowance of $1,000 in arrear, directly deposited into Sarah's bank account, with an interest rate of 5% p.a. compounded monthly....
-
Your supervisor, Viola Sylliboy of Dalcomm Financial Associates, has asked you to submit a progress report on your work on Aputik Prosper's Stock Performance and Recommendation Report. Because you...
-
Weight: 9% Submit: via the Dropbox tool Due: Friday of Week 10 by 11:59 pm ET Note: Please refer to the Outline for exact due dates. Choose arbitrarily one bond fund, one Canadian equity fund, and...
-
Tom Douglas is a famous Seattle restaurant entrepreneur. He has been so successful that he now just angel invests into new concepts that approach him. Recently a slew of entrepreneurs have reached...
-
You have been hired as a consultant by Steady-State Life Insurance Company. Steady-State specializes in the sale of Guaranteed Investment Contracts (GICs) which guarantee clients a specific dollar...
-
Choose an appropriate closed system and draw a bar diagram representing the energy conversions and transfers that occur during each process of Checkpoint 7.9: (a) a ball launching as the compressed...
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
Evaluate *x sin t t X lim x-3 x - 33 - dt
-
How would you synthesize racemic disparlure (Problem 18.46) from corn- pounds having ten or fewer carbons?
-
Treatment of 1, 1-diphenyl- 1, 2-epoxyethane with aqueous acid yields diphenylacetaldehyde as the major product. Propose a mechanism for the reaction. Ph H30+ PHCHCH Ph Ph
-
How would you prepare o-hydroxyphenyl-acetaldehyde from phenol? More than one step is required. HO o-Hydroxyphenylacetaldehyde CH-CO
-
1. What is the organ in the body that weighs just as much as the brain and is in some ways just as important to who we are? 2. What are some of the functions microbes perform in the body? 3. What...
-
In an engine, a piston oscillates with simple harmonic motion so that its position varies according to the expression x ( t ) = 15.0 cos( 20.0 t ) where x is in centimeters and t is in seconds. (a)...
-
When you think of your existence in the world, how do you think of it? How do you exist in the world? What is the world? What are you? What is the relationship between you and the world? Now consider...

Study smarter with the SolutionInn App


