Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H
Question:
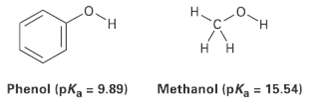
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use resonance structures to explain the difference in acidity.
Transcribed Image Text:
H. H. Methanol (pKa = 15.54) = 9.89) Phenol (pK
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
H 66666 HH When p...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Explain the following observations: (a) HNO3 is a stronger acid than HNO2; (b) H2S is a stronger acid than H2O; (c) H2SO4 is a stronger acid than HSO4-; (d) H2SO4 is a stronger acid than H2SeO4; (e)...
-
Explain the following observations: (a) HCl is a stronger acid than H2S; (b) H3PO4 is a stronger acid than H3AsO4; (c) HBrO3 is a stronger acid than HBrO2; (d) H2C2O4 is a stronger acid than HC2O4-;...
-
Use resonance formulas to explain why polyacetylene has delocalized molecular orbitals extending over the length of the molecule, whereas the following molecule does not. HHHHH
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
Samuel and Annamaria are married, file a joint return, and have three qualifying children. In 2016, they earn wages of $34,000 and no other income. Determine the amount of their earned income credit...
-
An aqueous glycerol solution weighing 100.0 mg was treated with 50.0 mL of 0.083 7 M Ce 4+ in 4 M HClO 4 at 60C for 15 min to oxidize the glycerol to formic acid: The excess Ce 4+ required 12.11 mL...
-
Which of the following statements about the single linkage method is false? a. In two dimensions, resulting clusters tend to be compact and circular. b. In two dimensions, the resulting clusters can...
-
On April 8, 2010, a flood destroyed the warehouse of Stuco Distributing Co. From the waterlogged records of the company, management was able to determine that the firms gross profit ratio had...
-
. Can you describe what's wrong with the below code? 1 testProc([1, 2, 3]) # Explicitly passing in a list 2 testProc() # Using a default empty list 3 4 def testProc(n = []): 5 # Do something with n 6...
-
QUESTION 1 ?Is this statement true or false??Even if plagiarism is discovered years from now, a university can withdraw a qualification that was awarded to a student? A. True B. False 1 points ?...
-
We?ll see that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristics chemical reactivity. Use...
-
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.
-
In 2005, Hurricane Katrina decimated the Gulf Coast of the United States between Mobile, Alabama, and New Orleans, Louisiana. The aftermath of this storm left the city of New Orleans flooded, both...
-
What is defined as the procedure used to standardize the instrument for accuracy by determining the instrument's deviation from calibration references (fine tuning)? What is defined as the procedure...
-
Stor Inc. owns 35% of the outstanding stock of Investee Company. During 2022, Investor received a $4,000 cash dividend which was declared and paid by the Investee. What effect did this have on...
-
Why are Reserves created by companies? Distinguish between (i) Free Reserves and 'Specific Reserve' and (ii) Revenue Reserve and Capital Reserve.
-
Maggie's Bar spent $113,800 to refurbish its current facility. The firm borrowed 75 percent of the refurbishment cost at 7.82 percent interest for six years. What is the amount of each monthly...
-
Whether for a corporate purchase or a personal one, the capital budgeting process is the same. For this assignment, you will analyze the investment (purchase) made for a new car, a decision most...
-
Discuss how Murimi can use her technical skills to devise the strategy. Renita Murimi is a currency overlay manager and market technician who serves institutional investors seeking to address...
-
Write a paper by answer the following question: Should Recycling Be Mandatory?
-
Using the Redlich-Kwong equation of state, compute the following quantities for nitrogen at 298.15 K. a. The difference C P C V as a function of pressure from low pressures to very high pressures b....
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Arrange these compounds in order of increasing SN1 reaction rate: Ph-Br Br + Br CI
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
4.5. Multiplication in GF(24): Compute A(x)-B(x) mod P(x) in GF(24) using the irreducible polynomial P(x) = x++x+1. What is the influence of the choice of the reduction polynomial on the computation?...
-
Write a C++ program that calculates the volume of a cylinder. Define appropriate variables for the diameter and the height of the cylinder. Assuming the units of these variables are in inches,...
-
a. Show the d and values that result from running breadth-first search on the directed graph of Fig 1, using vertex A as the source. (Ascending Order) b. Show the d, f and values that result from...

Study smarter with the SolutionInn App


