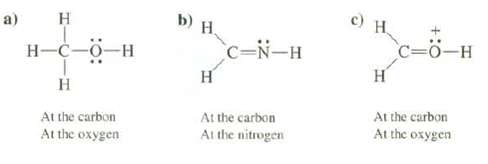
Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the
Question:
Predict the geometry of the following compounds at the indicated atoms:
Transcribed Image Text:
a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 92% (13 reviews)
a Tetrahedral at C bent at ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the geometry of the following molecules and ion using the VSEPR model: (a) CH3I, (b) ClF3, (c) H2S, (d) SO3, (e) SO42-?
-
Predict the geometry of the following ions, using the electron-pair repulsion model. a. ClO3 b. PO43 c. SCN d. H3O+
-
Predict the geometry of the following molecules using the VSEPR method: (a) HgBr2, (b) N2O (arrangement of atoms is NNO), (c) SCN- (arrangement of atoms is SCN)?
-
Which of the following options are available for creating a policy in Qualys Policy Compliance? (Choose three) A, Create from Host B, Create from Scratch C, Import from Library D, Import from CSV File
-
Monitor Ltd uses 1680 tonnes of a chemical each year in its manufacturing processes. Monthly demand for the chemical fluctuates between 110 and 160 tonnes. The lead time for each order is one month,...
-
The following data are the resting pulse rates for 30 randomly selected individuals who were participants at a 10K race. a. Construct a stem-and-leaf plot of the pulse rates. b. Construct a boxplot...
-
Distinguish between formulating statistical conclusions about the population and determining the acceptability of the population.
-
Annas, Inc., has a $350,000, 4 percent bond issue that was issued a number of years ago at face value. There are now 10 years left on the bond issue, and the market increase rate is 8 percent....
-
Present a 6-8 minutes persuasive speech persuading the audience about a social problem that our society needs to address. Use the problem-cause-solution organization to construct your argument and...
-
What volume of n-hexane is required to decrease the concentration of X in Problem 31-11 to 1.00 x 10-4 M if 25.0 mL of 0.0500 M X is extracted with (a) 25.0-mL portions? (b) 10.0-mL portions? (c)...
-
Predict the geometry at the carbon of these compounds: a) H-C=N: b) 0: H-C-0-H c) H T CI-C-H T CI
-
Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H H H-C-C=N H
-
Refer to figure 35.27 and the chapter content to answer the following questions. 1. Review the Survey the Landscape figure in the chapter introduction and the Pull It Together concept map. Explain...
-
Consider that you have recently joined as a design engineer at Ball Bearing Manufacturing Company in Gebang, Pahang, after graduating from Universiti Malaysia Pahang. Your task is to design an...
-
The ball has a mass of [m] kg. Let F= 300 N and d = 1 m. f) Determine the force TAB in cable AB. g) Determine the force Tac in cable AC. 1.5 m d B -2 m A D -F
-
Q2) A gear box is to be designed to reduce the input speed of 4800 rpm to 30 rpm. Available modules are 5mm, 8 mm and 10 mm and available pressure angles are 20 and 25. The gear box should be as...
-
b) A vibrating tuning fork is held next to an adjustable open-closed tube. The frequency of the tuning fork is also the fundamental frequency of the tube at its shortest length. When the tube slowly...
-
In the figure the lower disk, of mass 430 g and radius 3.4 cm, is rotating at 180 rpm on a frictionless shaft of negligible radius. The upper disk, of mass 260 g and radius 2.4 cm, is initially not...
-
During the period ended 30 June 2023, Ru Li, an employee of Westshore Company, purchased goods from the company on normal commercial terms and conditions. Li receives remuneration consisting of cash...
-
The Home Depot is the leading retailer in the home improvement industry and one of the 10largest retailers in the United States. The company included the following on its January 29, 2012, balance...
-
Find f g h if f(x) = x/(x + 1), g(x) = x 10 , and h(x) = x + 3.
-
Propose structures for molecules that meet the following descriptions: (a) Contains two sp2-hybridized carbons and two sp3-hybrized carbons (b) Contains only four carbons, all of which are...
-
Why cant molecules with the following formulas exist? (a) CH5 (b) C2H6N (c) C3H5Br2
-
Draw a three-dimensional representation of the oxygen-bearing carbons atom in ethanol, CH3CH2OH, using the standard convention of solid, wedged, and dashed lines.
-
1.Taxpayer, a cash method, calendar year taxpayer, engaged in the following transactions in shares of stock. Consider the amount and character of T's gain or loss in each transaction: (a)T bought 100...
-
where company shows it uneanedd revuene and how it is decribedd in the footnotes?
-
Required Compute variances for the following items and indicate whether each variance is favorable (F) or unfavorable (U): Note: Select "None" if there is no effect (i.e., zero variance). Item Sales...

Study smarter with the SolutionInn App


