Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H
Question:
Predict the direction of the dipole moments of these compounds:
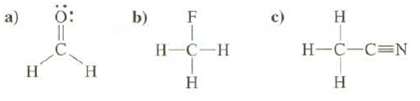
Transcribed Image Text:
a) H H b) F H-C-H H H H-C-C=N H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
First determine the geometry of the molecule by VSEPR theory Then f...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Use electronegativities to predict the direction of the dipole moments of the following bonds. (a) C-CI (b) C-O (c) C-N (d) C-S (e) C-B (f) NCl (g) NO (h) NS (i) NB (j) B-CI
-
Predict the direction of migration in an electrophoresis apparatus (toward the positive or negative electrode) of each component of the following amino acid mixtures: a. Glycine and lysine at pH 7 b....
-
The compounds FCl and ICl have dipole moments that are similar in magnitude (0.9 and 0.7 D, respectively) but opposite in direction. In one compound, chlorine is the positive end of the dipole; in...
-
Blaine is a practicing dentist. He operates his business from the basement of his house, with a separate entrance and facilities. Blaine uses the simplified method every year to calculate his...
-
Cascade of Poses (COP) is a business that specialises in the delivery of arrangements of roses. Cascade of Poses uses six main suppliers of roses, and has worked hard over the past two years to...
-
The regulations of the board of health in a particular state specify that the fluoride level must not exceed 1.5 parts per million (ppm). The 25 measurements given here represent the fluoride levels...
-
What factors should be considered by the auditor in analyzing occurrences found in the sample?
-
1. Prepare a list showing all tasks and their durations. 2. Analyze the fact situation carefully to determine which tasks are concurrent and which ones are dependant on other tasks. 3. Using PERT/CPM...
-
Reflect on this new information you have learned. Can you relate to your Sparketypes? Are they an accurate reflection of you? How will you apply what you have learned to working in groups this...
-
Design a cast iron protective flange coupling to connect two shafts in order to transmit 7.5 kW at 720 r.p.m. The following permissible stresses may be used : Permissible shear stress for shaft,...
-
Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
-
Explain whether the bonds in these compounds would be ionic or covalent and show Lewis structures for them: (a) KC1 (b) NC13 (c) NaCN (d) KOH
-
A test-marketing of a newly released DVD in a representative Canadian city, with a population of 120,000, resulted in sales of 543 units in a three-month period. If the DVD sells at the same rate in...
-
Jessica's team has been working on a new beverage product that she believes is almost ready for launch. However, the next step is to see how consumers react to the new beverage in a realistic...
-
A string of length L and total mass M is stretched to a tension T, as in figure 3. Tension is high enough that we can neglect gravity. 4 w/30* M L 4 w/30+ M 4 M 3 Figure 3: Sketch for Q3. 3.1. To...
-
Consider a world composed of two countries, Home (H) and Foreign (F). Individuals living in each country i = H, F have preferences over two goods and y. In each country there is only one factor of...
-
Two dimensions. In the figure, three point particles are fixed in place in an xy plane. Particle A has mass mA = 4 g, particle B has mass 2.00mA, and particle C has mass 3.00mA. A fourth particle D,...
-
1) The connecting rod AB of a certain internal combustion engine weighs 1.2 lb with mass center at G and has a radius of gyration about G of 1.25 in. The piston and the wrist pin (pin A) together...
-
Wilderness Windfarms is a government organisation which directly controls another entity Steam Energy Ltd. Through this investment it indirectly controls Blades Construction Ltd and Zepher Farms...
-
In the series connection below, what are the respective power consumptions of R, R2, and R3? R R www 4 V=6V P1-3 W; P2=3W; and P3= 3 W OP10.5 W; P2-1 W; and P3= 1.5 W P1=1.5 W; P2=1 W; and P3= 0.5 W...
-
Sketch the graph of the absolute value function f(x) = |x|.
-
Oxaloacetic acid, an important intermediate in food metabolism, has the formula C4H4O5 and contains three C = O bonds and two O H bonds. Propose two possible structures.
-
Draw structures for the following molecules (a) Acrylonitrile, C3H3N, which contains a carbon-carbon double bond and a carbon-nitrogen triple bond (b) Ethyl methyl ether, C3H8O, which contains an...
-
Potassium methoxide, KOCH3, contains both covalent and ionic bonds. Which do you think is which?
-
A tall pitot-static tube is mounted through and 1.5-mabove the roof of a performance car such that it senses thefreestream flow. Estimate the static, stagnation, and dynamicpressure sensed at 325 kph...
-
If cluster membership is determined by examining the squared distance of each respondent's position from the initial cluster centers, list the respondents assigned to initial Cluster#1 (5 points) ...
-
How does the perception of organizational justice affect employee motivation and job satisfaction, and what can organizations do to promote a sense of fairness and equity ?

Study smarter with the SolutionInn App


