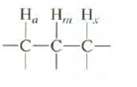
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain. , , . -C-C-C-
Question:
Predict the multiplicity of the absorption for Hm if Jam = Jmx. Explain.
Transcribed Image Text:
Н, Н, Н. -C-C-C-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
The signal from H m will appear ...View the full answer

Answered By

Pharashram rai
i am highly creative, resourceful and dedicated freelancer an excellent record of successful classroom presentations and writing . I have more than 4 years experience in tutoring students especially by using my note making strategies and engineering field . Especially adept at teaching methods of maths and writing , and flexible teaching style with the willingness to work beyond the call of duty. Committed to ongoing professional development and spreading the knowledge within myself to the blooming ones to make them fly with .
4.80+
65+ Reviews
270+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the multiplicity of each signal in the 1 H NMR spectrum of the following compound:
-
Predict the multiplicity (the number of peaks as a result of splitting) and the chemical shift for each shaded proton in the following compounds. (a) (b) (c) (d) (e) (f) CH3 CH3-C H-C H 3 CH CH3 CH 3
-
Predict the key IR absorption bands whose presence would allow each compound in pairs (a), (c), (d), (e), (g), and (i) from Problem 2.46 to be distinguished from each other. In problems 2.46 (a) (c)...
-
can you complete this concept map that reviews some key ideas about species and speciation logy with Lab Connecting the Concepts: Species and Speciation oncepts: Species and Speciation oncept map...
-
Discovered an error in computing a commission; received cash from the sales person for the over payment, $1000. Paid salaries and commissions for the month, $27,800 recorded revenue earned and billed...
-
Why do some consumers engage in out shopping? What could be done to encourage them to shop closer to home?
-
Find the z-score that has 88.9% of the distributions area to its right. Find the indicated z-score.
-
In its first tax year, the Vasquez Estate generated $50,000 of taxable interest income and $30,000 of tax-exempt interest income. It paid fiduciary fees of $8,000. The estate is subject to a 35%...
-
How have advancements in membrane technology impacted extraction processes? Explore the use of membrane-based methods like pervaporation and membrane distillation in the extraction and purification...
-
Wyoming Electric Coop owns a steam-turbine power-generating plant. Because Wyoming is rich in coal deposits; the plant generates its steam from coal. 111is, however, may result in emission that does...
-
Predict the approximate chemical shifts for the different hydrogen's in thesecompounds: CI CI a) CH,CH,CH3 b) CH;CHCH3 c) CH,COCH,CH3 d) CH;CHCH2
-
Construct a tree diagram for the absorption of Hm assume that Jam . . - -C-
-
Write the electron configuration for silicon. Identify the valence electrons in this configuration and the non valence electrons. From the standpoint of chemical reactivity, what is the important...
-
Company A results last year; S = $25,700; CGS = $18,800; D = $3,450; I = $990. Start of last year, NF = $19,280, CA = $5,100, and CL = $3,400, End of prior year NF = $23,650, CA =$5,830 and CL...
-
If the initial concentration and final concentration of each of the following components are given in the picture, how much volume in microliter (ul) is needed to go initial stock concentration to...
-
As a manager, you believe in the expectancy theory of motivation. which of the following actions would you take to influence the effort, performance, and reward perceptions that are integral to this...
-
What is Blackstone's theory of environmental rights?
-
Prior to 1960, the Coca-Cola Company produced only one soft drink aimed at the entire soft drink market. Since then, Coca-Cola has segmented the market based on diverse consumer preferences for...
-
How can you decide on the number of independent variables and that adding one more would improve the predictability of the dependent variable?
-
Suppose Green Network Energy needs to raise money to finance its new manufacturing facility, but their CFO does not think the company is financially capable of making the periodic interest payments...
-
Traditionally, manufacturers have relied on clay models to create three-dimensional images of what products on the drawing board might look like once they were constructed. Increasingly, however,...
-
Look at figure, and tell which molecular orbital is the HOMO and which is the LUMO for both ground and excited states of ethylene and 1, 3-butadiene. (b) Antibonding (3 nodes) Antibonding (2 nodes)...
-
Draw the products you would expect from conrotatory and disrotatory Cyclizations of (2Z, 4Z, 6Z)-2, 4, 6-octatrienc. Which of the Iwo paths would you expect the thermal reaction to follow?
-
Trans-3, 4-Dirnethvlcyclobutene can open by two conrotatory paths to give either (2E, 4E)-2, 4-hexadicne or (2Z, 4Z)-2, 4-hexadiene. Explain why both products are symmetry-allowed, and then account...
-
What is a plan on how you intend to be successful in future semesters. The plan should be a source of motivation to do well in college, both professionally and personally. Include tips that you can...
-
Question 1. From the research for this terms Crisis Management Team Assignment - Taylor swift and Ticket Master Crisis a. Explaining, with some detail, three (3) specific things that the leader(s) in...
-
If individual values that underline ethics are developed at a young age, what might this suggest about the potential for ethical conflicts to arise within an organization?

Study smarter with the SolutionInn App


