Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a
Question:
Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a mechanism for the following reaction of an ?, ? un-saturated ester.
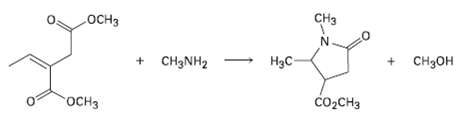
Transcribed Image Text:
CHз .ОCHЗ + CH3NH2 H3C- + CH3OH госнз CO2CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
CH3NH H3C CHO CH3O CHOH HC HC loss of methanol CH3NH HC conjugate addition of methy...View the full answer

Answered By

Sandhya Sharma
I hold M.Sc and M.Phil degrees in mathematics from CCS University, India and also have a MS degree in information management from Asian institute of technology, Bangkok, Thailand. I have worked at a international school in Bangkok as a IT teacher. Presently, I am working from home as a online Math/Statistics tutor. I have more than 10 years of online tutoring experience. My students have always excelled in their studies.
4.90+
119+ Reviews
214+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction: CH CH, C-CH, CH2OH Cl CH3
-
Propose a mechanism for the following reaction: CI Alcl
-
Propose a mechanism for the following reaction. (The rate of the reaction is much slower if the nitrogen atom is replaced by CH.) CI - H2o CH3 N CI
-
Read the case study about Joy Jowie Inc and write a detailed paper about it
-
1. As Social Enterprises try to get larger, what unique challenges do they face? 2. What are some of the drawbacks associated with partnering with governments or big businesses? If you ran an SE,...
-
During 2003, a share of stock in the Coca-Cola Company sold for $39. During 2008, the price hit $56 per share. Express the increase in price as a percent of the price in 2003. Round to the nearest...
-
What is the System Element Architecture (SEA) Model?
-
Holder-Webb Company began operations on January 1, 2005, and uses the average cost method of pricing inventory. Management is contemplating a change in inventory methods for 2008. The following...
-
Can a normal sized law firm handle this matter on our own or should they engage an Alternative Legal Service Provider? Why or why not? If the law frim was to engage an ALSP what kind of services...
-
The Excel file Cereal Data provides a variety of nutritional information about 67 cereals and their shelf location in a supermarket. Use regression analysis to find the best model that explains the...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 10 H 12 O ? ??IR: 1710 cm ?1 ? (b) C 6 H 12 O 3 ? ? ?IR: 1715 cm ?1 ? (c) C 4 H 6 O ? ? ? ?IR: 1690 cm ?1 ?...
-
When crystals of pure ?-glucose are dissolved in water, isomerization slowly occurs to produce ?-glucose. Propose a mechanism for the isomerization. CH2 CH2 - - B-Glucose a-Glucose
-
A tank having a volume of 0.100 m3 contains helium gas at 150 atm. How many balloons can the tank blow up if each filled balloon is a sphere 0.300 m in diameter at an absolute pressure of 1.20 atm?
-
New Center Co . has concluded a contract with the Defense Department for $ 4 9 5 , 0 0 0 with a forecasted cost of $ 3 7 5 , 0 0 0 . It is estimated at this time that 6 0 % of the job is complete...
-
A telephone manufacturer produced and sold 2 0 0 0 telephones and made a net income of $ 7 0 , 0 0 0 last year, with total revenues of $ 5 0 0 , 0 0 0 . The manufacturer s break - even volume is 1 2...
-
Stroud Corporation is an 80%-owned subsidiary of Pennie, Inc., acquired by Pennie several years ago. On January 1, 2017, Pennie sold land with a book value of $60,000 to Stroud for $90,000. Stroud...
-
Dozier Company produced and sold 1 , 0 0 0 units during its first month of operations. It reported the following costs and expenses for the month: Direct materials $ 7 2 , 0 0 0 Direct labor $ 3 6 ,...
-
[111] The inverse of the matrix A = 6 7 8 8 using 6 7-8 adjoint method is :
-
In Example 8.3 , we introduced a simple quantum system involving an electrically charged, spin- \(1 / 2\) particle immersed in a uniform magnetic field. In this exercise, we will continue the study...
-
Evenflow Power Co. is considering a new project that is a little riskier than the current operations of the company. Thus, management has decided to add an additional 1.5% to the company's overall...
-
Argon has a normal boiling point of 87.2 K and a melting point (at 1 atm) of 84.1 K. Its critical temperature is 150.8 K, and its critical pressure is 48.3 atm. It has a triple point at 83.7 K and...
-
Para-nitrosation of N,N-dimethylaniline (C-nitrosation) is believed to take place through an electrophilic attack by N+O ions. (a) Show how N+O ions might be formed in an aqueous solution of NaNO 2...
-
If we examine Table 21.1, we find that the methylphenols (cresols) are less acidic than phenol itself. For example, This behavior is characteristic of phenols bearing electron-releasing groups....
-
When o-chlorotoluene is subjected to the conditions used in the Dow process (i.e., aqueous NaOH at 350oC at high pressure), the products of the reaction are o-cresol and m-cresol. What does this...
-
Dewan INC. has several divisions, each with a manager responsible for the operations of the division. Each division of Dewan controls product design, sales, pricing, operating costs, and profits.....
-
Reed Company had the following account balances for the current year. Prepare a multiple-step income statement. U Accounts Receivable $ 26,000 Cost of Goods Sold 63,400 Dividends 22,900 General and...
-
Welch Corporation just paid its annual dividend of $2.00 per share. The firm is expected to grow at a rate of 15 percent for the next three years and then at 6 percent per year thereafter. The...

Study smarter with the SolutionInn App


