Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 10
Question:
Propose structures for ketones or aldehydes that have the following 1H NMR spectra:
(a) C10H12O ? ??IR: 1710 cm?1?
(b) C6H12O3? ? ?IR: 1715 cm?1?
(c) C4H6O ? ? ? ?IR: 1690 cm?1?
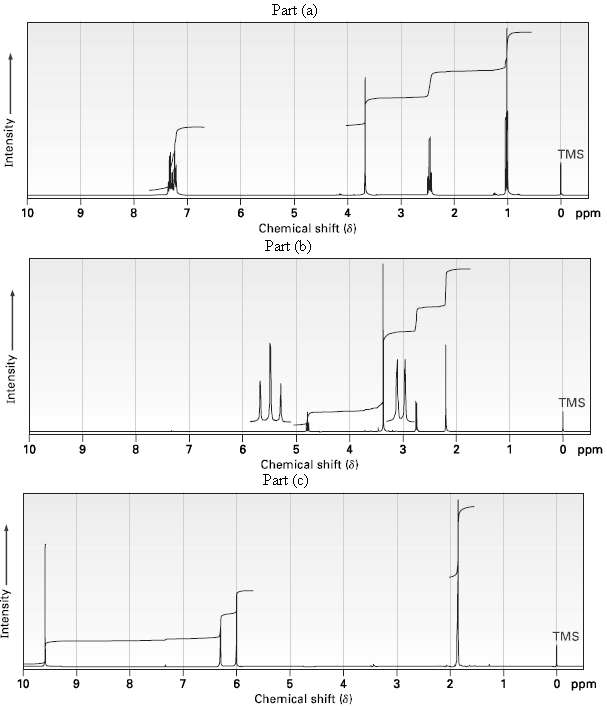
Transcribed Image Text:
Part (a) TMS 10 6. 5. Chemical shift (8) 3 0 ppm Part (b) TMS O ppm 10 9. 8. Chemical shift (8) Part (c) TMS 10 8. 4 O ppm Chemical shift (8) Intensity Intensity Intensity
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a CHCCHCH3 b a 108 b 258 C 3...View the full answer

Answered By

Muhammad Haroon
More than 3 years experience in teaching undergraduate and graduate level courses which includes Object Oriented Programming, Data Structures, Algorithms, Database Systems, Theory of Automata, Theory of Computation, Database Administration, Web Technologies etc.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
An airplane has a mass of 5000 kg, a maximum thrust of 7000 N, and a rectangular wing with aspect ratio 6.0. It takes off at sea level with a 60 split flap as in Fig. 7.25. Assume all lift and drag...
-
How would you describe Zappos as an organization? Mechanistic? Organic? High-or low-involvement? Why?
-
The GeoStar Company, a leading manufacturer of wireless communication devices, is considering three cost-reduction proposals in its batch job-shop manufacturing operations. The company has already...
-
By finding the trace of the appropriate coherency matrix, show that the average intensity transmitted by a quarter-wave plate followed by a polarization analyzer set at \(+45^{\circ}\) to the...
-
Dilberts Department Store is trying to determine how many Hanson T-shirts to order. Currently the shirts are sold for $21, but at later dates the shirts will be offered at a 10% discount, then a 20%...
-
A girl throws a rock horizontally with a speed of 12 m/s from a bridge above a river. The rock travels a horizontal distance of 27 m, before hitting the water. Neglect air resistance.(a) Find how...
-
An acoustic double-slit system (of slit separation d and slit width a) is driven by two loudspeakers as shown in Figure. By use of a variable delay line, the phase of one of the speakers may be...
-
Propose structures for ketones or aldehydes that have the following 1 H NMR spectra: (a) C 4 H 7 C1O ??IR: 1715 cm ?1 ? (b) C 7 H 14 O ? ? ?IR: 1710 cm ?1 ? (c) C 9 H 10 O 2 ? ? ?IR: 1695 cm ?1 ?...
-
Primary amines react with esters to yield amides: RCO2R' + R''NH2 ? RCONHR'' + R'OH. Propose a mechanism for the following reaction of an ?, ? un-saturated ester. CH .CH + CH3NH2 H3C- + CH3OH CO2CH3
-
Which of the following is not a payroll function? (a) Check gross pay entitlement of each employee (b) Verify accuracy of pay-sheet deductions (c) Prepare and distribute payslips (d) Notify...
-
What types of strategic choices do managers have when deciding on recruiting and selection efforts? How can the HR department and line manager collaborate to develop an authentic environment built...
-
Read a news report and an opinion piece on the same topic . step 2: write a one page summary on the topic . You can conduct this same experiment with any topic that interests you: contrast the...
-
Background on Valuation of Flow-Through Entities: The valuation of flow-through entities has been a debate since 1999 when the Tax Court decided an S corporation was more valuable than an otherwise...
-
How privacy laws impacts Amy and her employee's use of photographs taken for business clients ?
-
What are the three purposes that a bill of lading may have? How does an air waybill differ from a "traditional" bill of lading ?
-
Consider the model proposed in Daniel et al. [516] and described in Sect. 9.3, with uninformed agents and overconfident informed agents. Prove Proposition 9.12.
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Predict the shapes of the following molecules, and then predict which would have resultant dipole moments: (a) SO 2 ; (b) NH 3 ; (c) H 2 S; (d) C 2 H 4 ; (e) SF 6 ; (f) CH 2 Cl 2 .
-
Show how you might utilize the reduction of an amide, oxime, or nitrile to carry out each of the following transformations: (a) (b) (c) (d) NIH NH2 OH NH2
-
Using a different method for each part, but taking care in each case to select a good method, show how each of the following transformations might be accomplished: (a) (b) (c) (d) (e) NH2 NH2 CH3O...
-
Review the chemistry of amines given in earlier sections and provide a specific example for each of the previously illustrated reactions.
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?
Computer Aided Architectural Design Futures 2001 Volume I 1st Edition - ISBN: 0792370236 - Free Book

Study smarter with the SolutionInn App


