Propose a mechanism for the following reaction, an important step in the laboratory synthesis ofproteins: C=CH2
Question:
Propose a mechanism for the following reaction, an important step in the laboratory synthesis ofproteins:
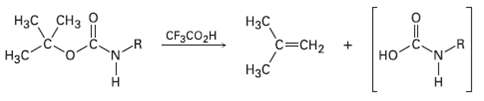
Transcribed Image Text:
Нас снз Нзс C=CH2 Нас CF3CO2H Нас но H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
H3C CH3 0 H3C CF3COH H3C H R CCH CF3CO...View the full answer

Answered By

Mamba Dedan
I am a computer scientist specializing in database management, OS, networking, and software development. I have a knack for database work, Operating systems, networking, and programming, I can give you the best solution on this without any hesitation. I have a knack in software development with key skills in UML diagrams, storyboarding, code development, software testing and implementation on several platforms.
4.90+
67+ Reviews
156+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Draw all missing reactants and/or products in the appropriate boxes by placing atoms on the canvas and connecting them with bonds. Add charges where needed. Electron flow arrows should start on the...
-
Propose a mechanism for the following transformation:
-
Burke Fabricators completed two jobs in June. Burke Fabricators recorded the following costs assigned to the jobs by the companys activity-based costing system: Job 622 required 2,400 parts, 77,500...
-
Given the information gathered from the selection methods, what process did Dolan use to make his selection decision? What improvements can you recommend to this process for decisions to hire sales...
-
Write a structural formula for each of the following: (a) 3-Methylcyclobutene (b) 1-Methylcyclopentene (c) 2, 3-Dimethyl-2-pentene (d) (Z)-3-Hexene (e) (E)-2-Pentene (f) 3, 3, 3-Tribromopropene (g)...
-
What conditions must an entity meet before recognizing revenue for financial statement reporting purposes?
-
Your firm designs PowerPoint slides for computer training classes, and you have just received a request to bid on a contract to produce the slides for an 8-session class. From previous experience,...
-
How collaborative leadership can help principals balance discipline with other responsibilities? How would you intentionally structure your leadership teams to address instructional leadership?
-
In August 2007 and in March 2008, the Federal Reserve Board reduced the discount rate to ease liquidity conditions for banks. Plot discount window borrowing (FRED code: DISCBORR) between January 2007...
-
SN2 reactions take place with inversion of configuration, and SN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of...
-
The amino acid methionine is formed by a methylthio reaction of homo?cysteine with N-methyltetrahydrofolate. The stereochemistry of the reaction has been probed by carrying out the transformation...
-
Simplify each of the following.
-
You have a convex lens with a focal length of 11.3cm. You want to use it to project an image from your cell phone's screen onto a wall that is 198cm away from the lens. How big the image would be on...
-
Three objects are set in motion, a 3.8 kg object at 1.6 m/s, a 2.7 kg object at 2.7 m/s, and a 1.6 kg object at 3.8 m/s. Which object has the most kinetic energy?
-
A volleyball is spiked so that its incoming velocity of +4.32 m/s is changed to an outgoing velocity of -19.0 m/s. The mass of the volleyball is 0.350 kg. What is the magnitude of the impulse that...
-
How do businesses navigate complex regulatory landscapes while ensuring compliance without compromising operational agility and innovation?
-
In what ways can businesses redesign their operations to be more customer-centric while maintaining cost efficiency and operational scalability?
-
What is the speed of an electron after being accelerated from INT rest through a \(20 \times 10^{6} \mathrm{~V}\) potential difference?
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
The reaction X 2 (g) 2 X(g) occurs in a closed reaction vessel at constant volume and temperature. Initially, the vessel contains only X 2 at a pressure of 1.55 atm. After the reaction reaches...
-
Show reaction sequences (not detailed mechanisms) that explain these transformations: (a) (b) CH,O 2 (1) NaOEt OEt 2) H COOH + CH2(COOEt)2 (1) NaOEt (2) HO+
-
Show how you would accomplish each conversion using an enamine synthesis with pyrrolidine as the secondary amine. (a) Cyclopentanone 2-allylcyclopentanone (b) Pentan-3-one...
-
Draw the mirror images of glucose and fructose. Are glucose and fructose chiral? Do you expect them to be optically active?
-
In this assignment, students must select a publicly listed entity, perform fundamental analysis and provide investment recommendations. The selected company can be one of the companies in your...
-
The following is an interesting interview with Ray Kurzweil that explains what the "Singularity" is. It is only two months ago, before the explosion Chat GPT....
-
When is a situation where a larger, more complex firm might have net income higher or lower than changes in cash (in other words, provide a concrete hypothetical setting where cash and net income...

Study smarter with the SolutionInn App


