SN2 reactions take place with inversion of configuration, and SN1 reactions take place with racemization. The following
Question:
SN2 reactions take place with inversion of configuration, and SN1 reactions take place with racemization. The following substitution reaction, however, occurs with complete retention of configuration. Propose amechanism.
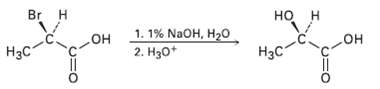
Transcribed Image Text:
но н Br H 1. 1% NaOH, H2O но" он 2. Hзо* он он H3C Нас
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Br H X CO OH NaOH Br O...View the full answer

Answered By

Krishna Kumar
I am a doctor. And i studied very hard during my degree period. But i believe that if you are giving proper time for outing and other your hobbies than you can done better in your study.
Study with fresh and relax mind give better results.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The phenomenon of configuration inversion in a chemical reaction was discovered in 1896 by Paul Walden (Section 6.6). Walden's proof of configuration inversion was based on the following cycle: (a)...
-
The following reaction occurs by a general-acid-catalyzed mechanism: Propose a mechanism for this reaction. CH2 CH3 HB+
-
The following reaction occurs by a mechanism involving general-base catalysis: Propose a mechanism for this reaction. base + CH,CH20H CH2OH
-
Mickey Limited is a manufacturing business that uses a standard costing system. The companys flexed budget for April 20X9 is: Sales 173,340 Costs Direct materials (22,500) Direct labour (37,684)...
-
How well does searching social media fulfill the requirements of providing reliable, valid, high-utility, and legal information for selection decisions?
-
Each of the following names is incorrect. Give the correct name and explain your reasoning. (a) trans-3-Pentene (b) 1, 1-Dimethylethene (c) 2-Methylcyclohexene (d) 4-Methylcyclobutene (e)...
-
What advantages do both franchisors and franchisees enjoy by entering into a franchise agreement, rather than conducting the entire scope of operations individually?
-
Cost of goods manufactured and sold The following information pertains to Pandey Manufacturing Company for March 2012. Assume actual overhead equaled applied overhead. Required a. Prepare a schedule...
-
How does the psychologic and economic impact differ from other forms of terrorism such as cyber terrorism? Also, how does a client's prejudices and attitudes can pose a danger to the personal...
-
Alpha Airlines has ordered a new fleet of DC- 717s. At this stage of the contract, Alpha's operations manager must specify the seating configuration on the aircraft that will be used on the...
-
When a primary alcohol is treated with p-toluenesulfonyl chloride at room temperature in the presence of an organic base such as pyridine, a tosylate is formed. When the same reaction is carried out...
-
Propose a mechanism for the following reaction, an important step in the laboratory synthesis ofproteins: C=CH2 CF3CO2H H.
-
Using everyday language rather than statistical language, explain why the word cutoff might have been chosen to define the point beyond which we reject the null hypothesis.
-
The Salad Oil Scandal of the early 1960s was one of the worst corporate scandals of its time. It occurred when executives at New Jersey-based Allied Crude Vegetable Oil Company discovered that banks...
-
Many public companies disclose alternative, non-GAAP earnings measures. Unlike GAAP measures such as operating income, earnings before taxes, or net income reported on the face of the income...
-
Annual credit sales of Nadak Company total $ 3 2 4 . 6 million. The firm gives a 2 . 2 5 % cash discount for payment within 1 0 days of the invoice date; 7 5 % of Nadak's accounts receivable are paid...
-
An airplane traveling at half the speed of sound emits a sound of frequency 4.14 kHz. (a) At what frequency does a stationary listener hear the sound as the plane approaches? kHz (b) At what...
-
A 40 kg child uses a pogo stick to bounce up and down. The spring constant, k , of the toy equals 8,650 N/m. (a) By how much would the spring be compressed by the child if she simply balanced herself...
-
The highest-energy cosmic ray ever detected had an energy of about \(3.0 \times 10^{20} \mathrm{eV}\). Assume that this cosmic ray was a proton. a. What was the proton's speed as a fraction of \(c\)...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Consider the reaction for the decomposition of hydrogen disulfide: A 0.500-L reaction vessel initially contains 0.0125 mol of H 2 S at 800 C. Find the equilibrium concentrations of H 2 and S 2 . 2...
-
A reaction involved in the metabolism of sugars is the splitting of fructose-1,6-diphosphate to give glyceraldehyde-3-phosphate and dihydroxyacetone phosphate. In the living system, this retro-aldol...
-
Give the expected products of the following acid-catalyzed reactions. (a) Acetophenone + methylamine (b) Acetophenone + dimethylamine (c) Cyclohexanone + aniline (d) Cyclohexanone + piperidine
-
Biochemists studying the structure of collagen (a fibrous protein in connective tissue) found cross-links containing ,-unsaturated aldehydes between protein chains. Show the structures of the side...
-
Should an MNC purchase Nestl stock yes or no? Recommendations should include the company's sustainability plans, the image of the company, Gordon Growth result, and financial performance. Any...
-
Can the sponsor, or general partner, continue to achieve such high returns? Framed another way, can the general partner earn a 25+% return on the $6,305,000 received at the end of the 10th year? If...
-
Select a hospital that publicly displays their financial performance on their website. Describe the Profit Loss Statement: Review top revenue items Review cost comparison compared to previous year...

Study smarter with the SolutionInn App


