Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra: 100 80
Question:
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra:
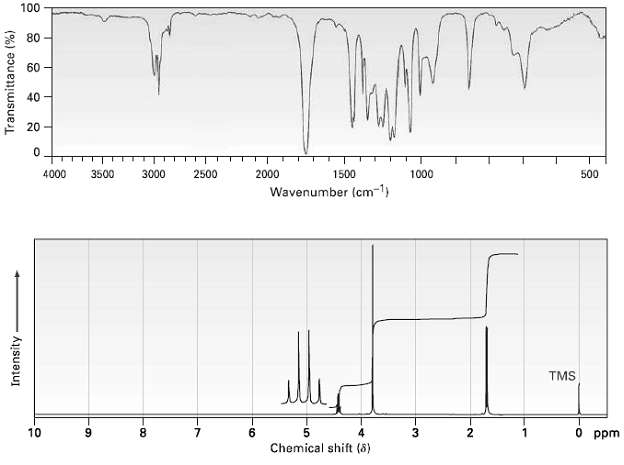
Transcribed Image Text:
100 80 60 40 20 4000 3000 2500 2000 1000 500 3500 1500 Wavenumber (cm-1) TMS O ppm 10 6. Chemical shift (8) Transmittance (%) Intensity -08- -LO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
The IR spectrum indic...View the full answer

Answered By

Chiranjib Thakur
I have no tutoring experience yet, but I can share my skills and knowledge gained from my education and work experiences. I have been a CPA since 2012 with 6 years of work experience in internal auditing and 4 years of work experience in accounting at the supervisory level.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a structure for a compound with molecular formula C 10 H 14 O that exhibits the following 1 H NMR spectrum. Proton NMR Chemical shift (ppm)
-
Propose a structure for a compound with molecular formula C 3 H 8 O that exhibits the following 1 H NMR and 13C NMR spectra: Proton NMR 0.5 5.0 4.5 4.0 3.5 3.0 2.0 1.5 1.0 25 Chemical shift (ppm)...
-
Propose a structure for a compound with molecular formula C 8 H 10 O that exhibits the following 1 H NMR spectrum: Proton NMR Chemical shift (ppm)
-
IKEA was founded in 1943 by a 17-year-old Swede named Ingvar Kamprad. The company, which initially sold pens, Christmas cards, and seeds from a shed on Kamprad?s family farm, eventually grew into a...
-
How do report writers decide what type of graphic to use in a report?
-
Plants outside the plot have mean seed number 15.0. Consider again the weights, heights, yields, and seed number for 10 plants given in Exercises 1-4. Find the sample variance for each and use the t...
-
Can semantic networks be used to represent anything that can be represented using temporal logic? Explain your answer.
-
Osborne Manufacturing is preparing its master budget for the first quarter of the upcoming year. The following data pertain to Osborne Manufacturings operations: Current assets as of December 31...
-
Assuming an arbitrage-free world, today you buy a zero-coupon bond with $100 par value and 1.5 years to maturity, and your holding period is six months. Based on the information of question 21, you...
-
Suppose that, in their divorce settlement, Ashton Kutcher offers Demi Moore $10 million spread evenly over 10 years, but she instead demands $5 million now. If the appropriate discount rate is 8...
-
How would you distinguish spectroscopically between the following isomer pairs? Tell what differences you would expect to see. (a) N-Methylpropanamide and N, N-dimethylacet amide (b)...
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
The income statement and additional data of Floral World, Inc., follow: Additional data: a. Collections from customers are $30,000 more than sales. b. Payments to suppliers are $1,000 more than the...
-
1. The running time for the following code fragment is (f(n)). What is f(n)? for (int i=0; i
-
b) What is the output of the following code fragment? int arr [10] = {-2, 6, 9, -1}; int *pl, j; p1 = arr; p1++; p1++; for (j = 1; j
-
In 2015 Medicaid total enrollment was 82.5 million persons. Spending by state and federal government on Medicaid was $552.3 billion. Of the different groups of persons enrolled which group was the...
-
What unit of measure would be used to measure factors (patient, healthcare workers, emergency department setting/environment) that influence violence against healthcare workers in the emergency...
-
How many companies believe they offer equal pay regardless of gender? What is unconscious bias? What did researchers observe in a Stanford University study? How many STEM jobs will go unfilled due to...
-
Is the following rock a sedimentary rock, igneous rock, or metamorphic rock? Why do you think so? Sand grains Cement Pore space (open space filled with water)
-
Rewrite Programming Exercise 7.5 using streams. Display the numbers in increasing order. Data from Programming Exercise 7.5 Write a program that reads in 10 numbers and displays the number of...
-
What is an antibonding molecular orbital?
-
Using the information in Table 16.2, predict the product(s) of Table 16.2 Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of A1C13. Eq. 16.23 TABLE 16.2...
-
Using the information in Table 16.2, predict the product(s) of Table 16.2 Friedel-Crafts alkylation of a large excess of ethylbenzene with chloromethane in the presence of A1C13. Eq. 16.23 TABLE 16.2...
-
Draw the carbocation that results from the reaction of the electrophile at the ortho position of anisole; show that this ion also has four resonance structures.
-
Despite Warren East's restructuring, Rolls-Royce still has a 'tall' organisational structure. Two options for the future of Rolls-Royce are to remain as a tall structure or to change to a flat...
-
Discuss and compare the strategies of a key competitor to Ulta Beauty. Discuss important differences and similarities in strategy. What does the competitor's future look like?
-
Coca-Cola introducing a new line of chai tea is an example of which market-product strategy?

Study smarter with the SolutionInn App


