Propose a structure for a molecule C 14 H 12 that has the following 1 H NMR
Question:
Propose a structure for a molecule C14H12 that has the following 1H NMR spectrum and has IR absorptions at 700, 740, and 890 cm?1:
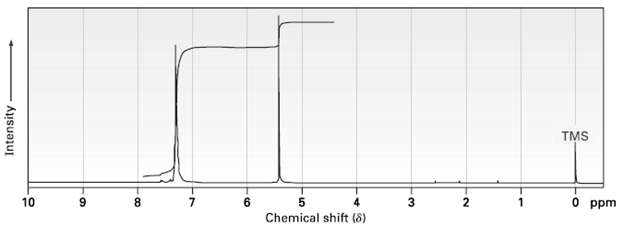
Transcribed Image Text:
TMS 3 5 Chemical shift (8) 10 O ppm 8 7 Intensity-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
The compound has nine degrees of unsaturation The H NMR ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Propose a structure for an alcohol with molecular formula C5H12 O that has the 1H NMR spectrum given in Fig. 9.46. Assign the chemical shifts and splitting patterns to specific aspects of the...
-
Propose a structure for a compound with molecular formula C 4 H 8 O that exhibits the following 13 C NMR and FTIR spectra. Carbon NMR 67.7- 25.4- 80 30 20 100 90 70 60 40 10 Chemical shift (ppm) 100...
-
Private carriage is more important in the motor carrier segment of our transportation industry than any of the other four major modal segments. What factors have contributed to private carriage...
-
How do the basic human rights - free consent, privacy, freedom of conscience, freedom of speech, and due process - apply to professional football players and the safety risks described here?
-
The beta of Tasty Foods stock of 1.10 used by Rae in computing the required return on equity was based on monthly returns for the past 10 years. If Rae uses daily returns for the past 5 years, the...
-
For each of the following situations, calculate the standard error of the mean \(s \mathrm{X}^{-}\). a. \(s=5.00 ; N=16\) b. \(s=17.82 ; N=10\) c. \(s=2.31 ; N=37\) d. \(s=51.32 ; N=21\)
-
Young, Martinez, and Cheung (YMC) is a law firm in Chicago. The firm has had a very loose and relaxed management style that has served it well in the past. However, more aggressive law firms have...
-
What is the output? int main() { for (int i = 0; i < 3; ++i) { cout < < i; } } cout < < i; return 0;
-
Forecasting with the Parsimonious Method and Estimating Share Value Using the ROPI Model Following are income statements and balance sheets for Cisco Systems. Cisco Systems Consolidated Statements of...
-
Propose structures for aromatic compounds that have the following 1 H NMR spectra: (a) C 8 H 9 Br IR: 820 cm ?1 (b) C 9 H 12 Br IR: 750 cm ?1 (b) C 11 H 16 Br IR: 820 cm ?1 TMS 10 8. 6. 0 ppm...
-
Aromatic substitution reactions occur by addition of an electrophile such as Br+ to the aromatic ring to yield an allylic carbocation intermediate, followed by loss of fl. Show the structure of the...
-
Marsellus Manufacturing incurred the following costs in acquiring land, making land improvements, and constructing and furnishing a new building. a. Purchase price of four acres of...
-
Critically critique your reading assignment for this module and point out any types of bias you may discern. Identify what type of bias and modify the statement(s) to show how you would present the...
-
Finally, in column J compare your company's ratios to the industry's ratios. Enter a 'U' if your firm's ratios compare unfavorably with industry standards. Enter an 'F' if your firm's ratios compare...
-
Felix is working for a marketing firm making $60,000 per year but considers starting his own marketing company. Felix has determined that to launch the business, he needs to invest $100,000 of his...
-
A global corporation that fabricated financial records to increase sales and mislead investors was implicated in a recent accounting controversy. This unlawful behavior was revealed by a...
-
What are environmental storage conditions for storing for: 1. tonkotsu ramen stock 2. pho stock 3. oxtail soup (kkori gomtang) 4 .veg stock(clear vegetable soup) 5. hot and sour soup 6. miso soup 7....
-
Let \(\mathbf{X}\) be a \(d\)-dimensional random vector with distribution function \(F\). Let \(g: \mathbb{R}^{d} ightarrow \mathbb{R}\) be a continuous function such that \(|g(\mathbf{x})| \leq b\)...
-
The text defined intrinsic value as the value of an asset given a hypothetically complete understanding of the assets investment characteristics. Discuss why hypothetically is included in the...
-
Consider the reaction: A(g) 2 B(g). The graph plots the concentrations of A and B as a function of time at a constant temperature. What is the equilibrium constant for this reaction at this...
-
Oils containing highly unsaturated acids like linolenic acid undergo oxidation in air. This reaction, called oxidative rancidity, is accelerated by heat, explaining why saturated fats are preferred...
-
Give equations to show the reactions of sodium stearate with (a) Ca2+ (b) Mg2+ (c) Fe3+
-
Several commercial laundry soaps contain water-softening agents, usually sodium carbonate (Na2CO3) or sodium phosphate (Na3PO4 or Na2HPO4). Explain how these water-softening agents allow soaps to be...
-
Can social norms be understood as emergent properties of social interactions, reflecting negotiated agreements among members of a society regarding acceptable behavior and moral standards?
-
A quantitative portfolio manager is very good at managing against the Russell 2000 Index. In fact, she is expected to achieve an B of 0.5% per month. Unfortunately, her benchmark is the S&P 500. She...
-
Suppose that a QEPM department hires a new quantitative equity portfolio manager to manage one of its portfolios. Suppose that the portfolio managers benchmark is the S&P 500, with a Sharpe ratio of...

Study smarter with the SolutionInn App


