Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS
Question:
Propose structures for alcohols that have the following 1H NMR spectra:
(a) C9H12O
(b)C8H10O2
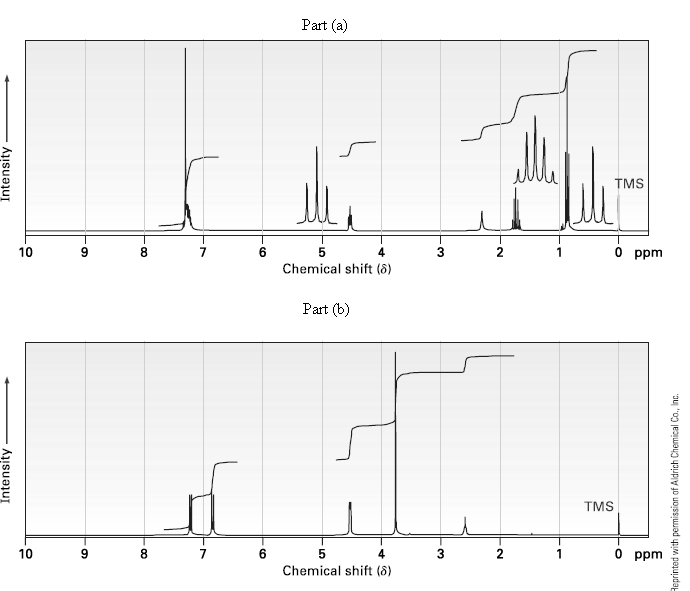
Transcribed Image Text:
Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity 3. 3. Reprinted with permission of Aldrich Chemical Co., Inc.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
a e OH C CHCHCH3 d ba 1Phen...View the full answer

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
In the Tokyo subway system, routes are labeled by letters and stops by numbers, such as G-8 or A-3. Stations allowing transfers are sets of stops. Find a Tokyo subway map on the web, develop a simple...
-
How can a mission statement be an enduring statement of values and simultaneously provide a basis of competitive advantage?
-
Refer to Exercise 14.27. a. Use Tukeys multiple comparison method to determine which pairs of political parties differ. b. Use an appropriate method the compare the three Independents with the other....
-
Consider the simple linear regression model fit to the toluene-tetralin viscosity data in Problem 2.15. Problem 2.15 Byers and Williams ("Viscosities of Binary and Ternary Mixtures of Polynomatic...
-
The following letter was sent to the SEC and the FASB by leaders of the business community. Dear Sirs: The FASB has been struggling with accounting for derivatives and hedging for many years. The...
-
Espresso Express operates a number of espresso coffee stands in busy suburban malls. The fixed weekly expense of a coffee stand is $ 1 , 3 0 0 and the variable cost per cup of coffee served is $ 0 ....
-
Tom Jackson was an employee of a large farm machinery manufacturing plant located in Akron, Ohio. He had been recently promoted and was considered by his peers to be a very good employee. Toms new...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
Refer to the facts in Exercise 11-39. In Exercise, Douglas manufactures four grades of lubricant, W-10, W-20, W-30, and W-40, from a joint process. Additional information follows: Required Assuming...
-
International macroeconomics question a) Why do economists typically view trade as beneficial to all countries involved? In your answer, explain what are the gains to trade. Are all individuals...
-
Is the paternity procedure begun in California with a complaint, a petition, or some other pleading?
-
In Apple Inc. How well does the business model capture the organization's unique value proposition? Does it clearly define how the organization delivers value to its customers? Is the target market...
-
Congress recently enacted the Family Legal Services for the Poor Act. The Act establishes a Poverty Law Office in every state that is responsible for representing indigent clients in family law...
-
A shopping cart filled with food ( m = 4 0 kg ) is pushed along the floor with uk = 0 . 3 and moves with an acceleration of 0 . 3 m / s ^ 2 . What horizontal force was being exerted on the cart?
-
True or false; explain your answer: If all teams are of equal quality, it doesnt matter whether they share gate receiptsrevenue will remain unchanged.
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
Water molecules will form small, stable clusters. Draw one possible water cluster by using six water molecules and maximizing the number of hydrogen bonds for each water molecule.
-
Referring to the retrosynthetic analysis for 2-methylhexane in this section, write reactions for those synthesis routes that are feasible
-
(a) Devise retrosynthetic schemes for all conceivable alkynide anion alkylation syntheses insect pheromones undecane and 2-methylheptadecane. (b) Write reactions for two feasible syntheses of each...
-
Each of the following names is incorrect. Give the correct name and explain your reasoning. (a) trans-3-Pentene (b) 1, 1-Dimethylethene (c) 2-Methylcyclohexene (d) 4-Methylcyclobutene (e)...
-
Let 1 f(z) = (2-1)(x-2) Find the Laurent expansion of f about z = 0 in the region {ze C: 1
-
Find all the complex numbers z such that z += 0. Is the set open or closed? {zC: z*+i=0}
-
For each of the following matrices A Maxn (R), test A for diagonal- izability, and if A is diagonalizable, find an invertible matrix Q and a diagonal matrix D such that Q-1AQ = D. (a) (63) 2 1 3 (b)...

Study smarter with the SolutionInn App


