Propose a mechanism to account for the following reaction: C CH2CI AICI3
Question:
Propose a mechanism to account for the following reaction:
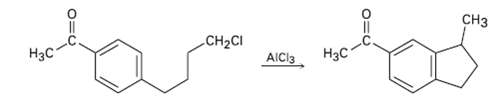
Transcribed Image Text:
Cнз CH2CI Нас Нзс AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
H CHCl love formation of H3C0 H3C HC CIAICI3 CH3 HA loss of H AICI 3 CH3 ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Propose a mechanism to account for the fact that heating 1-deuterioindene scrambles the isotope label to all three positions on the five-memberedring. 1-Deuterioindene
-
What is the result of the following? A. [Natural History, Science] B. [Natural History, Science, Art] C. The code does not compile. D. The code compiles but throws an exception at runtime. } import...
-
Morgan (age 45) is single and provides more than 50% of the support of Rosalyn (a family friend), Flo (a niece, age 18), and Jerold (a nephew, age 18). Both Rosalyn and Flo live with Morgan, but...
-
Medical billing errors and fraud are on the rise. According to Medical Billing Advocates of America, 8 out of 10 times, the medical bills that you get are not right. (Data extracted from "Services...
-
What steps should be taken by the auditor after he has received a cutoff statement?
-
The annual report of General Mills, maker of Wheaties, Cheerios, and Betty Crocker baking products, for the year ended May 29, 2011, contained the following ($ in millions): During fiscal 2011,...
-
Simplify the following switching functions. The simplified result should be in SOP format. Show all work the way I show in class. Don't take short cuts. Note: The book does not make this limitation....
-
1. In 2020, Orasco Sa had net sales of 600,000 and cost of goods sold of 360,000. Operating expenses were 153,000 and interest expense was 7,500. The corporations tax rate is 30%. The corporation...
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl 3 gives...
-
Lisa Lasher buys 400 shares of stock on margin at $18 per share. If the margin requirement is 50 percent, how much must the stock rise for her to realize a 25 percent return on her invested funds?...
-
The Sendai Framework for Disaster Risk Reduction (UNDRR, 2015) calls for investments in digital technologies and tools to enhance societal resilience. Recent developments in digital technologies and...
-
In a multidisciplinary group of therapists, the group member who most often helps to oversee group decision - making and actions would best fit which role from Benne and Sheats' typology?
-
Consider in a market where the supply and demand functions for a product are given as follows: Supply function : P S = 20 + 2Q Demand function : P D = 50 - Q where P S is price given by supplier, P D...
-
You observe that the inflation rate in Canada is 2.4% per year and that T-bills currently yield 2.9% annually. a. What do you estimate the inflation rate to be in Australia, if short-term Australian...
-
If actual inflation rate is 8% and expected inflation rate is 6.5%, what is unemployment rate?
-
Nestls financial statements are presented in Appendix B. Financial statements of Delfi Limited are presented in Appendix C. Instructions Refer to the financial statements and answer the following...
-
Anna, a high school counselor, devised a program that integrates classroom learning with vocational training to help adolescents at risk for school dropouts stay in school and transition to work...
-
Electrical power is to be produced from a steam turbine connected to a nuclear reactor. Steam is obtained from the reactor at 540 K and 36 bar, the turbine exit pressure is 1.0 bar, and the turbine...
-
Show the resonance structures for the conjugate base of phenol.
-
The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic acid (H2CO3) is 6.35. Complete these equations and predict whether the reactants or the products are favored...
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
Question 1: How do strategy and tactical action relate to each other in an organization? Question 2: How can you improve the implementation of strategy in this business organization?...
-
Larkin Corporation sells products for $125 each that have variable costs of $50 per unit. Larkin's annual fixed costs are $430,000. (10 points Total) a. What is Larkin's contribution margin per unit?...
-
Find the determinant of the linear transformation T(M): [3] M from the space V of 22 upper triangular matrices to V.

Study smarter with the SolutionInn App


