Propose structures for compounds that show the following 1H NMR spectra. (a) C9H13N (b)C15H17N TMS 10 6
Question:
Propose structures for compounds that show the following 1H NMR spectra.
(a) C9H13N
(b)C15H17N
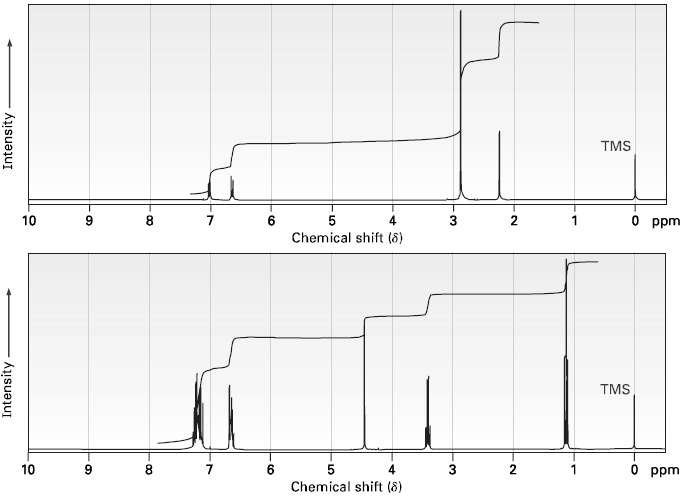
Transcribed Image Text:
TMS 10 6 5 3 2 O ppm Chemical shift (8) TMS 3 2 O ppm 10 6 1 Chemical shift (8) Intensity Intensity-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a HC NCH32 a 238 b ...View the full answer

Answered By

David Ngaruiya
i am a smart worker who concentrates on the content according to my clients' specifications and requirements.
4.50+
7+ Reviews
19+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following 1H NMR spectra are for four compounds with molecular formula C6H12O2, Identify the compounds. a. b. c. QUESTION CONTINUE TO NEXT PAGE d. 10 (ppm) frequency 10 (ppm) 10 (ppm)
-
Propose structures for compounds that fit the following 1 H NMR data: (a) C 4 H 6 Cl 2 2.18 (3 H, singlet) 4.16 (2 H, doublet, J = 7 Hz) 5.71 (1 H, triplet, I = 7 Hz) (b) C 10 H 14 1.30 (9 H,...
-
Propose structures for compounds X, Y, and Z: The 1H NMR spectrum of X gives two signals, a multiplet at δ 7.3 (5H) and a singlet at δ 4.25 (2H); the 680-840-cm-1 region of...
-
Remove left recursion in the following grammar. Show each step. Hint: First remove direct left recursion. Then the indirect left recursion. A AB | Aab | BA| a Bb | | b
-
Assume that you achieved all the tasks shown in Activity 4, not as an individual employee but as part of a work team. In your résumé, must you mention other team members? Explain your...
-
Describe how to prepare 100 mL of 0.200 M acetate buffer, pH 5.00, starting with pure liquid acetic acid and solutions containing ~3 M HCl and ~3 M NaOH.
-
What types of audits are addressed in the GAO's Government Auditing Standards'?
-
In a particular month Northwest Medical Clinic reported the following: 1. It provided direct care services to patients, billing them $400,000. Of this amount it received $120,000 in cash, but as a...
-
Setup: You are in a car on a North-South roadway with no bends, no other cars, and no intersections. You can put the car in drive (Northbound direction) or reverse (Southbound direction), and you can...
-
Alexander Smith and his wife Allison are married and file a joint tax return for 2019. The Smiths live at 1234 Buena Vista Drive, Orlando, FL 32830. Alexander is a commuter airline pilot but took 6...
-
Propose structures for amines with the following 1H NMR spectra: (a) C3H9NO (b)C4H11NO2 TMS O ppm 10 6. Chemical shift (8) TMS O ppm 10 7. 9. Chemical shift (8) Intensity Intensity -3-
-
?-Amino acids can be prepared by the Strecker synthesis, a two-step process in which an aldehyde is treated with ammonium cyanide followed by hydrolysis of the amino nitrile intermediate with aqueous...
-
What trade offs do buffer trees pose as compared to LSM trees?
-
Assume Ikeo Inc. sold $100,000 of gift cards during the last two weeks of December of Year 1. No gift cards were redeemed in Year 1. A total of $90,000 of the gift cards were redeemed for store...
-
Question: Which taxpayer may file as head of household? ( If the scenario states the dependent is a qualifying child or qualifying relative, it means they have passed all the dependency requirements....
-
In the current year, Brutus sold a non-depreciable $1231 asset at a gain of $7,000. He has an unrecaptured $1231 loss from three years earlier of $4,000. Assuming Brutus has no other sale...
-
What is a "contingent asset?" O There is no such thing, in IASB standards, as a "contingent asset." This is an asset that has been put up as collateral against a loan. This is a possible inflow of...
-
Question 3: I a) What is the 'recognition issue' in financial reporting? (5 marks) b) The energy and commodities company Enron aggressively used 'Mark-to-market' accounting policies. Research this...
-
Choose a publicly held corporation and find the balance sheet and notes to the financial statements in the most recent Form 10-K. This form can be located by going to the home page of the Securities...
-
Economic feasibility is an important guideline in designing cost accounting systems. Do you agree? Explain.
-
Determine if the solution formed by each salt is acidic, basic, or neutral. (a) SrCl 2 (b) AlBr 3 (c) CH 3 NH 3 NO 3 (d) NaCHO 2 (e) NH 4 F
-
There are three dibromobenzenes (o-, m-, and p-). Suppose we have samples of each in separate bottles, but we don't know which is which. Let us call them A, B, and C. On nitration, compound A (mp...
-
Give the structure and name of each of the following aromatic hydrocarbons: a. C8H10; has two possible ring-substituted monobromo derivatives b. C9H12; can give only one mononitro product after...
-
The observed amount of heat evolved when 1,3,5,7-cyclooctatetraene is hydrogenated is 110 kcal/mol. What does this tell you about the possible resonance energy of this compound?
-
Popular furniture company, IKEA, has purchased forests in Romania as well as land in Alabama to assist with keeping up with the wood demand necessary to complete customer orders. This was one way...
-
How does China being Turkey's biggest import partner affect Turkey's exchange rate?
-
Assignment 4 In this assignment you are provided information on an experiment and you are required to investigate and interpret the output which is provided below. Problem: Consider the...

Study smarter with the SolutionInn App


