A proton with velocity v = 107 m/s enters a region of uniform magnetic field B =
Question:
A proton with velocity v = 107 m/s enters a region of uniform magnetic field B = 0.8 T, which is into the page, as shown in Figure. The angle ? = 60o. Find the angle ? and the distance d.
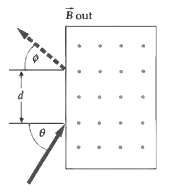
Transcribed Image Text:
B out
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (18 reviews)
The trajectory of the particle is shown From s...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
A particle of charge q enters a region of uniform magnetic field B (pointing into the page). The field deflects the particle a distance d above the original line of right, as shown in Fig. 5.8. Is...
-
As shown in Figure P18.42, water is pumped into a tall vertical cylinder at a volume flow rate R. The radius of the cylinder is r, and at the open top of the cylinder a tuning fork is vibrating with...
-
As shown in Figure P29.72, a particle of mass m having positive charge q is initially traveling with velocity vj . At the origin of coordinates it enters a region between y = 0 and y = h containing a...
-
Why do countries respond differently in terms of flexibility and worklife balance to what seem similar economic pressures?
-
1. A taxpayer's spouse died on December 31, 2014. He has no qualifying child. Which status should the taxpayer select when filing his 2015 tax return? a. Qualifying widow(er). b. Married filing...
-
The homogeneous circular cylinder of mass m and radius R carries a slender rod of mass m/2 attached to it as shown. If the cylinder rolls on the surface without slipping with a velocity v O of its...
-
During the assembly of an exhaust valve, sufficient distance must be maintained between the valve tip and the cylinder wall. If \(85 \%\) of valve assemblies have the required distance, use Table 1...
-
The following information, in T-account format, is provided for Mars Company for the year 2012: Additional information: a. Sales revenue for the period was $164,000. Accounts receivable (net)...
-
What are the advantages and disadvantages of organizing a business as a corporation? 2. How does a partnership differ from a limited liability company? 3. Why do corporates file for bankruptcy? 4....
-
The US Womens Swimming Team won the 1500 m relay at the 2016 Olympic Games. Here are the completion times, in seconds, for all eight teams that competed in the finals: a. Calculate the sample...
-
A beam of particles with velocity v enters a region of uniform magnetic field B that makes a small angle with v. Show that after a particle moves a distance 2 (m/qB)v cos measured along the...
-
Suppose that in Figure B = 0.6 T, the distance d = 0.4 m, and ? = 24o. Find the speed v and the angle ? if the particles are (a) Protons and (b) Deuterons Bout
-
In an adiabatic process, energy can be exchanged in the form of_________ .
-
What is the output of the following? A. highlights B. Newsweek C. The code does not compile. D. The code compiles but throws an exception at runtime. class Magazine implements Comparable { private...
-
How many of the following are legal declarations? A. None B. One C. Two D. Three E. Four. [] String lions = new String[]; String[] tigers = new String[1] {"tiger"}; - String bears [] = new String[]...
-
What is the output of the following application? A. ABC B. ABD C. ABC followed by a stack trace D. ABD followed by a stack trace E. AD followed by a stack trace F. None of the above. package game;...
-
What is the result of the following? A. The code does not compile. B. Line 14 throws an exception. C. Line 15 throws an exception. D. Line 16 throws an exception. E. Line 17 throws an exception. F....
-
What is the result of the following? A. 2 3 B. 2 56 C. 3 3 D. 3 56 E. None of the above. var list = new ArrayList (); list.add(56); list.add(56); list.add (3); var set = new TreeSet (list);...
-
Take the information you have gathered and your assessment of needs so far and suggest a system to meet the departments current needs, as well as future needs and opportunities. Prepare a paper that...
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
What specific control problems exist in developmental psychology with (a) Crosssectional studies (b) Longitudinal studies?
-
An ideal gas has a molar heat capacity Cv at constant volume. Find the molar heat capacity of this gas as a function of its volume V, if the gas undergoes the following process: (a) T = Toe v ; (b) p...
-
One mole of an ideal gas whose adiabatic exponent equals undergoes a process p = P0 + a/V, where P0 and a are positive constants. Find: (a) Heat capacity of the gas as a function of its volume; (b)...
-
One mole of an ideal gas with heat capacity at constant pressure Cp undergoes the process T = To + aV, where To and a are constants. Find: (a) Heat capacity of the gas as a function of its volume;...
-
do not use chatgpt or any other ai tool. A monopolist with a linear demand curve will have a marginal revenue curve with intercept and slope as the demand curve..
-
7. Consider the figure below. HO -C pka-COOH-2.19 pka-NH2 = 9.67 pka-sidechain 4.25 CHCH2C OH a. What amino acid is this? (1) b. Is it in the R or S configuration? (2) c. Draw the three forms of the...
-
2. Draw the structure of the missing major organic product(s) or reactant(s) in each of the transformations below including showing stereochemistry when appropriate. For reactions that produce an...

Study smarter with the SolutionInn App


