Rank the anions within each series in order of increasing basicity, lowest first. Explain. CH(CH2) CH CH
Question:
Rank the anions within each series in order of increasing basicity, lowest first. Explain.
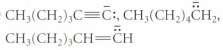
Transcribed Image Text:
CH(CH2) CH CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Because alkynes are more acidic than alkenes wh...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following compounds in order of increasing basicity. CH OH NH
-
Arrange the following in order of increasing basicity: (a) (b) (c)
-
Within each series arrange the compounds in order of increasing stability: C(CH3)3 HO CH,CH(CH)
-
Given the monthly returns that follow, find the R2, alpha, and beta of the portfolio. Compute the average return differential with and without sign. Do not round intermediate calculations. Round your...
-
What is the most dangerous job at the company?
-
In the network shown, the power absorbed by R is 5 mW. Find R. 1 R. 2
-
Superheated steam at \(250 \mathrm{psia}\) and \(500^{\circ} \mathrm{F}\) is compressed to \(350 \mathrm{psia}\). The isentropic efficiency of the compressor is \(70 \%\). For the compressor, compute...
-
Oahe Enterprises was formed by the efforts of Emmick, who acted as a promoter and contributed shares of Colonial Manors, Inc. (CM), stock in exchange for stock in Oahe. The CM stock had been valued...
-
You are an expatriate (with a spouse and a 6-year-old daughter) sent on assignment for one year to Hong Kong. What should you do to prepare for departure? Why? What training should you or your family...
-
Air FranceKLM (AF), a Franco-Dutch company, prepares its financial statements according to International Financial Reporting Standards. AFs financial statements and disclosure notes for the year...
-
On the basis of the hybrid orbitals involved in the bonds, arrange the bonds in each of the following sets in order of increasing length. C--C single bond of propan; C--C single bond of propyne; C--C...
-
Using simple observations or chemical tests with readily observable results, show how you would distinguish between the compounds in each of the following pairs. (a) l-hexyne and 2-hexyne (b) propyne...
-
ROCKAUTO.COM is a leading online auto parts store that prides itself for offering a huge selection of auto parts, everyday low prices, fast shipping, and an easy-to-use Web site. Some ROCKAUTO.COM...
-
OBJECTIVE: The objective of this assignment is to: Analyze business letters for effective use of information. Use the correct words in communicating ideas and information. Thus, you will develop your...
-
(a) (b) (c) Consider the following dataset. Gender Car Type Shirt Size Class label M M M M F F F M M M Family Sports Sports Sports Sports Sports M F F F Luxury Family Family Family Luxury Luxury...
-
Angle between two planes is defined to be the angle between their normals which 0 0 < (see the satisfies figure). Consider two planes with equations 2y + z = 14 and 2x + z = 2. For these planes...
-
3. For the mechanisms shown below, (1) Find the locations of all instant centers. (2) Given the angular velocity of Link 2 ,-10 rad/s(CW), Find the linear velocity of Point (Vc). The radius of the...
-
Louis Viggio was doing something he had never done before, creating a cash budget.This process was something his banker had asked him to do before coming in to talk to her about hiscurrent line of...
-
A random selection of 1000 smartphone owners, ages 18-24 years old, were asked if they constantly check and use their phone. A total of 518 responded yes. (a) Estimate the probability that, a...
-
What is beacon marketing? What are digital wallets?
-
Some peptides found in nature have an amino-terminal acetyl group (red): (a) Can these peptides undergo the Edman degradation? Explain. (b) Does N-acylation have any adverse effect on sequencing by...
-
(a) Give the m/z values of the fragment ions expected from b-type fragmentation of an M + 1 ion of the peptide N-F-E-S-G-K. (b) Give the m/z values of the fragment ions expected from y-type...
-
When a peptide Q with the amino acid analysis (A,F,G 2 ,I,K,N,P,R,S,Y) is treated with trypsin, three new peptides are formed (amino acid analysis in parentheses): T1(A,F,R,S); T2(G,I,K); and...
-
Choose from the following topics to create discussion: Humanity and the Natural Environment Housing and Home Furnishings Privacy and Socializing
-
On December 31, 2020, Pearl Company had $1,101,000 of short-term debt in the for January 21, 2021, the company issued 22,800 shares of its common stock for $42 per brokerage fees and other costs of...
-
During your interview process for the position of program manager for Booker, Tee, and How you are asked to explain the relationship between cost, schedule, and performance? what is your answer to...

Study smarter with the SolutionInn App


