Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76)
Question:
Rank the following substances in order of increasing acidity:
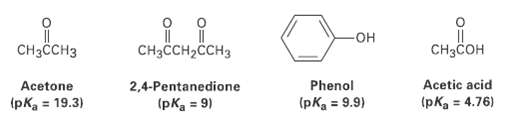
Transcribed Image Text:
Cнзсон CH3CH3 CHзCCH2CCHз Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (14 reviews)
The substances with ...View the full answer

Answered By

Jayshree Rathi
Hello Students!
This is Jayshree Rathi. I work on a number of renowned student-centric channels such as Chegg, coursehero, as a certified private tutor.
If you are looking for relevant and original content to complete your assignments, essays, and homework, then contact me and within the promised time, I will deliver you your personalized academic work and help you score the best.
4.80+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC CH, (CF3)2CHOH, CH4OH (b) Phenol, p-methyl phenol, p-(trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxy...
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following substances in order of their expected SN1 reactivity: Br Br H CHCCH CHCH2Br H2C=CHBr
-
Prepare adjusting journal entries, as needed, considering the account balances excerpted from the unadjusted trial balance and the adjustment data. A. depreciation on buildings and equipment, $17,500...
-
Individuals who receive substantial Social Security benefits are usually not eligible for the tax credit for the elderly or disabled because these benefits effectively eliminate the base upon which...
-
The space shuttle's expendable booster engines derive their power from solid reactants: 6NH4+ ClO-4 (s) + 10Al(s) 3N2(g) + 9H2O(g) + 5Al2O3(s) + 6HCI(g) (a) Find the oxidation numbers of the...
-
Leggere, an Internet book retailer, is interested in better understanding the purchase decisions of its customers. For a set of 1,604 customer transactions, it has categorized the individual book...
-
You are auditing a bank, and someone provides you with an anonymous tip that an employee is embezzling money from the bank. You decide to investigate the allegation. Your interviews with other bank...
-
This week, you will learn a relativist approach to ethics and examine how people make decisions based on groups, cultures and affiliations they belong to and how this shapes decision-making. You will...
-
The Valley Swim Club has 300 stockholders, each holding one share of stock in the club. A share of club stock allows the shareholders family to use the clubs heated outdoor pool during the summer,...
-
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain - C-OH - C=C c=C ...
-
Which, if any, of the four substances in Problem 2.43 is a strong enough acid to react almost completely with NaOH? (The pKa of H2O is 15.74.)
-
The plate has a mass of 3 kg and is welded to the fixed vertical shaft, which rotates at the constant speed of 20 rad/s. Compute the moment M applied to the shaft by the plate due to dynamic...
-
A firm which named Beyonce is currently growing fast. Current risk free rate is 7%; however, since the firm is somewhat risky investors requires a risk premium. So the required rate of return for the...
-
Explain the terms required in Texas for an enforceable physician's noncompete provision.
-
Explain how each of these 4 strengths listed below, will enable a person to develop a healthy organisational compliance culture or aid in this development. The answer must provide an excellent...
-
A water supply system was designed to provide water for the domestic use of a small community from a reservoir with the surface elevation of the reservoir at 300 ft. (Neglect minor losse) Pipe A and...
-
How would a manager use economic theory to determine profit-maximizing price for a service or product? What is the process of target costing and how is target cost calculated?
-
Based on Exhibit 2, the currency overlay program most appropriate for Braunt Pensionskasse would: A. be fully passive. B. allow limited directional views. C. actively manage foreign exchange as an...
-
Write a paper about the Working relationship in the organization- collaboration within and outside the organization
-
Redo Problem 4.45 if ethylene is described by the Peng-Robinson equation of state. Problem 4.45 The second virial coefficient B can be obtained from experimental PVT data or from an equation of state...
-
Assign the configurations of the compounds represented by these Fischer projections as R or S. CH -CH2CH3 b) CH2=CH- ) >N- CH-OH CH3 Serine
-
Explain whether each of these compounds is chiral or not: a) d) H C H c=c=c CHCH CCH HC PhwSi-OCH-CH b) CHCH Ph-N-CHCH, HC Br H C CH30 CH CO H c) f) H H HC CH30 CCH CH3 COH CH3 CO H
-
Although this biphenyl is chiral, it cannot be resolved. Explain. HO-C CH30 CO H OCH
-
You are the owner of a very small business that sells gourmet coffee. You sell only one product, a 12-ounce bag of whole-bean French roast coffee. You sell each bag of coffee for $14 each, but due to...
-
Find the instantaneous rate of change of the function f(t)=3+5t when t=-3.
-
Pretend you need to conduct a title search. You have the information needed from your online search, but you need to go to the clerk's office to finish your search. What information/records will/can...

Study smarter with the SolutionInn App


