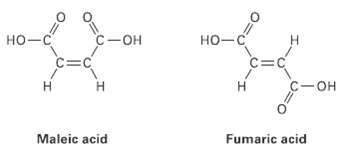
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the
Question:
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain
Transcribed Image Text:
с — он но-с C-OH но-с C=C c=C С —он Maleic acid Fumaric acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
In maleic acid the individual dipole moments ad...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A water molecule has a dipole moment of 6.3 x 10 30 C m. A sample contains 1021 molecules of water. Their dipole moments are all oriented in the direction of an electric field of 2.5 x 10 N/C....
-
The PF3 molecule has a dipole moment of 1.03 D, but BF3 has a dipole moment of zero. How can you explain the difference?
-
The ammonia molecule (NH3) has a dipole moment of 5.0 X 10-30 C m. Ammonia molecules in the gas phase are placed in a uniform electric field E with magnitude 1.6 X 106 N/c. (a) What is the change in...
-
A bank reconciliation takes time and must balance. An employee was struggling in balancing the bank reconciliation. Her supervisor told her to plug (make an unsupported entry for) the difference,...
-
Elaborate rules exist that require employers to prepay various types of Federal taxes. Summarize the major issues that an employer must resolve if it is to comply with the requirements?
-
Given the following information, find Ka for nitrous acid, HNO2. NO-3 + 3H+ + 2e- HNO2 + H2O Eo = 0.940 V E = 0.433 V
-
Freelance reporter Irwin Fletcher is examining the historical voting records of members of the U.S. Congress. For 175 representatives, Irwin has collected the voting record (yes or no) on 16 pieces...
-
"Sanders Enterprises, Inc., has been considering the purchase of a new manufacturing facility for $164,500. The facility is to be fully depreciated on a straight-line basis over 7 years. It is...
-
Explain how quality pedagogical practice is reinforced by marte meo and research about children's learning?
-
QUESTION 1 ?Is this statement true or false??Even if plagiarism is discovered years from now, a university can withdraw a qualification that was awarded to a student? A. True B. False 1 points ?...
-
Assign formal charger to the atoms in each of the followingmolecules: ( NNEN: CH (c) HCN3DN-DN: (a) H3C-N-O: CH
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
How does a product manager manage a products life cycle?
-
Our primary job's gross income is $3,468.73/month. Deductions are FICA (7.65%), federal tax withholding (11.5%), and state tax withholding (7.8%). How much are your fixed expenses if they are 36% of...
-
Why would an organization deliberately overstate their expenses during the current year?
-
I NEED HELP COMING UP WITH OPEN ENDED QUESTIONS MY RESEARCH ON POSITIVE PSYCHOLOGY As a Qualitative researcher who will be conducting interviews on your topic, write out some open-ended questions...
-
What are some useful ways in which you can demonstrate that you would be an asset to the company?
-
What 8 developments prior to 1960 were important to the development of blood evidence?
-
The active currency management approach that Umlauf Management is least likely to employ is based on: A. volatility trading. B. technical analysis. C. economic fundamentals. Guten Investments GmbH,...
-
Continuation of Exercise 4-83. (a) What is the probability that the first major crack occurs between 12 and 15 miles of the start of inspection? (b) What is the probability that there are no major...
-
a. Show that b. Use the result of part (a) to show that for a stable system at equilibrium ( V /T) S and ( V /T) P must have opposite signs. c. Two separate measurements are to be performed on a gas...
-
Draw all of the stereo isomers of 1, 2-dimethylcyclopropane. Explain which rotate plane-polarized light.
-
Explain whether these compounds rotate plane-polarized light: H a) HC-CCH, HC Br c) Br J. s CI b) Cl CI d) HC -CH3
-
Draw Fischer projections for these compounds? CHOH a) H-C-CI CH3 COH b) HC-OH A CH3 c) H CHCH CH CH3
-
Royal Lawncare Company produces and sells two packaged products-Weedban and Greengrow. Revenue and cost Information relating to the products follow: Selling price per unit Variable expenses per unit...
-
This is a slip and fall case. Plaintiff was injured when she slipped and fell in the defendant's fast food restaurant. Plaintiff claims the floor was wet and slippery when she fell. Defendant denies...
-
1. Explain what the "Deinstitutionalization Mandate" is. This should be done in at least two paragraphs. 2. In another two paragraphs, discuss how status offenses are connected to the...

Study smarter with the SolutionInn App


