Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
Question:
Rank the following substituted phenols in order of increasing acidity, and explain your answer:
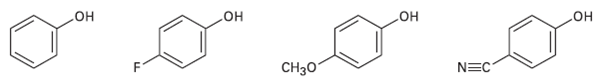
Transcribed Image Text:
CHзо но но но он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Remember that electronwithdrawing groups stabilize phenox...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following complex ions in order of increasing wavelength of light absorbed. Co(H20+ CoCN)-, Col, Colen)
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following five compounds in order of increasing acid strength: 2-chloroethanol, p-chlorophenol, p-methylphenol, ethanol, and phenol.
-
Which of the following is not an element of the fraud triangle? (a) Rationalization. (b) Financial pressure. (c) Segregation of duties. (d) Opportunity.
-
How does environmental analysis at the domestic level differ from global analysis?
-
Deprival value removes significant disadvantages of replacement cost, while retaining its advantages. Discuss.
-
The town of Commuter Park operates a private parking lot near the railroad station for the benefit of town residents. The guard on duty issues annual prenumbered parking stickers to residents who...
-
General Long-term Liability and Capital Asset Transactions The Village of Nassau issued a 3-year, 6 percent note in the amount of $100,000 to finance the purchase of vehicles for the Public Works...
-
Write a C++ program that calculates the total number of days between two dates (inclusive), which are input by the user, in a format of day month year. For example, if the input is 1 1 2022 and 11 4...
-
In this mini-case, you will complete the test of details on accounts receivable for the 2019 audit of EarthWear Clothiers, Inc. The principal test of detail involves sending "confirmations" or...
-
Benzoquinone is an excellent dienophile in the DielsAlder reaction. What product would you expect from reaction of Benzoquinone with 1 equivalent of 1, 3-butadiene from reaction with 2 equivalents of...
-
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the...
-
The CPA firm of Bigelow, Barton, and Brown was expanding rapidly. Consequently, it hired several junior accountants, including a man named Small. The partners of the firm eventually became...
-
Reply this experiences, explaining your ideas about how the situation could have been improved by the service provider or the company. Also answer the following in your REPLY: 1. Could a proactive...
-
The following data are available for PFE Inc. for July 2023. Selling price $45 per unit Direct Materials $250,000 Direct Labor $175,000 Manufacturing overhead $200,000 Selling expense $4.75 per unit...
-
Larned Corporation recorded the following transactions for the just completed month. a. $87,000 in raw materials were purchased on account. b. $85,000 in raw materials were used in production. Of...
-
You may hear someone say, "Technology makes people lazy." Do you think this is true? Evaluate the impact that changes in technology have had on your personal growth and development. For example, did...
-
Toby wants to calculate her COGS for a fertilizing job she did last week. She used 70 pounds of fertilizer that she bought in bulk ( $ 1 , 0 0 0 for 2 , 0 0 0 pounds ) . It took her employee 1 . 5...
-
Wyatt Ltd. is unable to reconcile the bank balance at January 31. Wyatts reconciliation is as follows. Instructions a. Prepare a correct bank reconciliation. b. Journalize the entries required by the...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
Show that the formation of NaCl 2 (s) is very unfavorable; that is, fH[NaCl 2 (s)] is a large positive quantity. Assume that the lattice energy for NaCl 2 would be about the same as that of MgCl 2 ,...
-
Classify the following solvents as being protic or aprotic: formic acid, HCO2H; acetone, CH3COCH3; acetonitrile, CH3CN; formamide, HCONH2; sulfur dioxide, SO2; ammonia, NH3; trimethylamine, N(CH3)3;...
-
Would you expect the reaction of propyl bromide with sodium cyanide (NaCN), that is, CH3CH2CH2Br + NaCN CH3CH2CH2CN + NaBr to occur faster in DMF or in ethanol? Explain your answer.
-
When tert-butyl bromide undergoes solvolysis in a mixture of methanol and water, the rate of solvolysis (measured by the rate at which bromide ions form in the mixture) increases when the percentage...
-
Mark Twain popularized the phrase "There are three kinds of lies: lies, damned lies, and statistics." How do people manipulate data and statistics to deceive? Give two examples. Provide at least...
-
As illustrated opposite, a rope passing through a system of pulleys C, D, E is attached at B to a rod OA of length L - 10cm and mass m = 1500g connected to a pivot at O. A mass m1 suspended from...
-
What are some managerial implications or consequences of ethical leadership at a company? 2. What is ethical leadership and what is it's impact on an organization? How might authoritative and...

Study smarter with the SolutionInn App


