Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves
Question:
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the anion with benzyl chloride and subsequent E2 reaction. Write the mechanism in detail, using curved arrows to indicate the electron flow in each step.
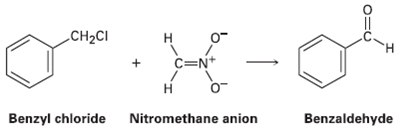
Transcribed Image Text:
CH2CI н C=N+ Н Benzyl chloride Nitromethane anion Benzaldehyde
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
09100 CH 0 ...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using curved arrows to indicate the election flow in each step, show how the base-catalyzed reverse aldol reaction of 4-hydroxy-4-methly-2-pentanone takes place to yield 2 equivalents of acetone.
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Show how 4-methylcyclohexyl chloride can be converted to 4- methylcyclohexanecarboxylic acid.
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
The model below depicts industry analysis as a funnel that focuses on remote-factor analysis to better understand the impact of factors in the operating environment. Do you find this model...
-
To carefully prepared mitochondria were added succinate, oxidized cytochrome c, ADP, orthophosphate, and sodium cyanide. Referring to Figure 14.16, answer the following. (a) List the sequence of...
-
To the right of z = 2.62 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
An intangible asset cost $300,000 on January 1, 2011. On January 1, 2012, the asset was evaluated to determine whether it was impaired. As of January 1, 2012, the asset was expected to generate...
-
3. A racecar with a mass of 1000 kg is traveling around a circular track with a radius of 50 meters at a speed of 100 km/h. What force is required to keep the car moving in a circle?
-
Claytonhill Beverages Ltd. is 100 percent owned by Buzz Bottling. While the company has in the past been profitable, it incurred a loss for the year ended December 31, 2012. The parent company, Buzz...
-
Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
-
Reduction of 2-butanone with NaBH4 yields 2-butanol. Is the product chiral is it optically active? Explain.
-
The specific rotations of pure a- and -d-fructofuranose are 121 and 2133, respectively. Solutions of each isomer mutarotate to an equilibrium specific rotation of 292. Assuming that no other forms...
-
How much is the petty cash fund shortage at December 31, 2021? The adjusted petty cash fund balance of Lannister Corporation at December 31, 2021 is? What is the amount of operating expenses found in...
-
The U.S. Department of Transportation reported the accompanying data on the number of speeding-related crash fatalities during holiday periods for the years from 1994 to 2003. Holiday Period 1994...
-
IV. ACME Corp. prepared the flexible budget items based on 1700 meals for a period. Actually 1800 meals are served during the period and financial records are given below. Evaluate the company's...
-
A computer-controlled ball launcher on a ledge at height h = 5 m above level ground ejects a ball at initial speed vo = 6.4 m/s at an angle of 0 = 38 degrees above the horizontal. Assume that air...
-
Davis and North, partners in an advertising business, share profits and losses in the ratio of 7:4, respectively. Prior to recording the admission of Martin as a new partner, Davis has a capital...
-
Discuss the major threats to validity associated with this design and type of research (experimental). Develop a hypothetical research scenario that would necessitate the use of an A-B-A Design. The...
-
Refer to the information from Exercise 22-19. Use the information to determine the (1) Weighted average contribution margin , (2) Break-even point in units, and (3) Number of units of each product...
-
The enthalpy of formation of NaI(s) is -288 kJ mol -1 . Use this value, together with other data in the text, to calculate the lattice energy of NaI(s).
-
List the following compounds in order of decreasing reactivity toward CH3O- in an SN2 reaction carried out in CH3OH: CH3F, CH3Cl, CH3Br, CH3I, CH3OSO2CF3, 14CH3OH.
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Which alkyl halide would you expect to react more rapidly by an SN2 mechanism? Explain your answer. (a) (b) (c) (d) (e) Br r or or CI
-
According to the Hawail Wildlife Fund, North Pacific Humpback Whales migrate from the icy waters around Alaska during the fall to spend the winter in Hawaii where they mate, give birth, and nurture...
-
Find the limit 4 lim (x -2x32x + x 1) =? 2+1 -
-
Train-the-trainer manual for the training and development department within your organization or for one with which you are familiar. Your trainers will soon be responsible to train the staff about...

Study smarter with the SolutionInn App


