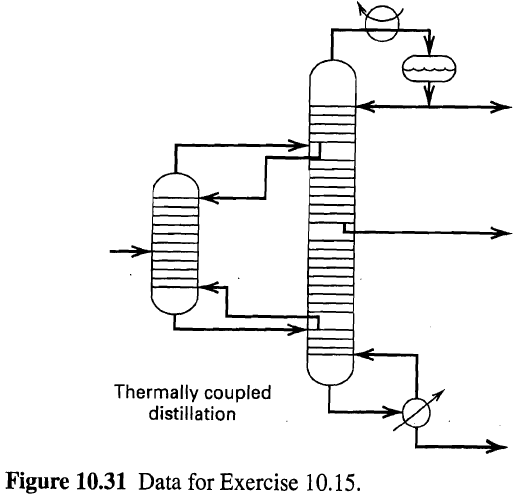
Revise equations (10-58) to (10-60) to allow two inter- linked columns of the type shown in Figure
Question:
Revise equations (10-58) to (10-60) to allow two inter- linked columns of the type shown in Figure to be solved simultaneously by the NR method. Does the matrix equation that results from the Newton'Raphson procedure still have a block tridiagonalstructure?
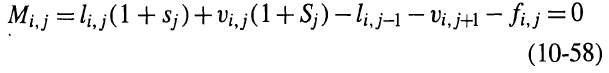
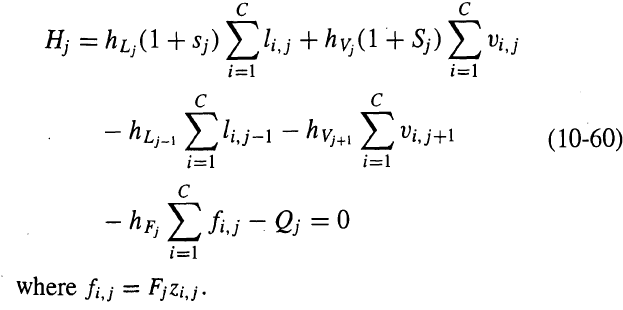
Transcribed Image Text:
Mij =lij(1+ 8;)+ vi, j(1+S;)– li, j-1 – vi, j+l – fi.j =0 (10-58)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Consider the system shown below where the stages in the first column are n...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
A coffee-cup calorimeter of the type shown in Figure 5.18 contains 150.0 g of water at 25.1 C. A 121.0-g block of copper metal is heated to 100.4 C by putting it in a beaker of boiling water. The...
-
A monolithic membrane element, of the type shown in figure d, contains 19 flow channels that are 0.5 cm in inside diameter by 0.85 m long. If nine of these elements are placed into a cylindrical...
-
In a microscope of the type shown in Figure the focal length of the objective is 4.00cm and that of the eyepiece is 8.00 cm. The distance between the lenses is 25.0 cm. (a) What is the tube length s?...
-
On 1 May 2006, Nigel acquired a 30-year lease for 20,000. He assigned the lease on 1 November 2020 for 75,000. Kay purchased land in February 2012 for 20,000. She sold one-third of the land for...
-
Financial statements aim to provide reliable information to a diverse group of users. Information must be enough for internal and external users to make decisions, but not so much that a company...
-
Consider the applecomputer trade example given in Section 19.5. Now suppose that because of a new technology, the North becomes even more productive at producing computers: one unit of labor can now...
-
Tip Top Advertising, Inc., engaged in the following business transactions during July of 2010: Tip Top Advertising, Inc., uses the following accounts: Cash, Accounts Receivable, Supplies, Building,...
-
Refer to the information for Deporte Company above. Deporte Company produces single-colored t-shirts. Materials for the shirts are dyed in large vats. After dying the materials for a given color, the...
-
For the past four years as a regular employee of the company, your performance rating has always been "outstanding". However, during the start of the current year, a new manager joined the company...
-
Ellen asks you to give her a list of the books that each author wrote. Write a complete SQL statement to provide the information to Ellen, including author names, book titles, publishers, and the...
-
A rigorous partial NR method for multicomponent, multistage vapor-liquid separations can be devised that is midway between the complexity of the BP/SR methods on the one other and the NR methods on...
-
In Equation (10-63) why is the variable order selected as v, T, l? What would be the consequence of changing the order to l, v, T? In Equation (10-64) why is the function order selected as E, M, H?...
-
Complete the following statements with one of the terms listed here. You may use a term more than once, and some terms may not be used at all. a. ________ cannot be directly traced to a (n) ________....
-
75. A mass m is attached to a spring which is held stretched a distance x by a force F, Fig. 6-45, and then released. The spring pulls the mass to the left, towards its natural equilibrium length....
-
It seems straight line depreciation was used above to calculate the depreciation expense- many businesses choose this method. What other methods can be used? Make a case as a management leader for...
-
A company had the following assets and liabilities at the beginning and end of this year. Beginning of the year End of the year Assets $ 142,500 190,000 Liabilities $ 61,090 76,950 a. Owner made no...
-
125. Ubisch bodies are produced by which layer of microsporangia :- (1) Middle layer (2) Epidermis (3) Endothecium (4) Tapetum 126. In the flower of Mussaenda, advertising flag is modification of?...
-
What % asset allocation should one set up for oneself assuming they have just graduated? Explain why u picked what u did.
-
When the expansion follows the law \(P V^{n}=C\), the process is: (a) Isothermal process (b) Adiabatic process (c) Polytropic process (d) Hyperbolic process
-
A woman at a point A on the shore of a circular lake with radius 2 mi wants to arrive at the point C diametrically opposite on the other side of the lake in the shortest possible A time. She can walk...
-
Explain why the General Electric Capital Corporation bond in the Cracking the Code box on p. 290 is selling at a premium above par?
-
For a batch rectifier with appreciable column holdup: (a) Why is the charge to the still-pot higher in the light component than at the start of rectification, assuming that total-reflux conditions...
-
One hundred lbmol of benzene (B), monochlorobenzene (MCB), and o-dichlorobenzene (DCB) is distilled in a batch rectifier that has a total condenser, a column with 10 theoretical stages, and a...
-
One hundred lbmol of 10 mol% propane, 30 mol% n-butane, 10 mol% n-pentane, and the balance n-hexane is to be separated in a batch rectifier equipped with a still-pot, a total condenser with a liquid...
-
You have just retired with savings of $ 1 . 5 million. If you expect to live for 3 0 years and to earn 8 % a year on your savings, how much can you afford to spend each month? Assume that you spend...
-
Mrs . Page had just completed a series of dental appointments. Typically Mrs . Page is a positive and very complimentary of the dental healthcare team. However, today you notice that she is very...
-
what is my principal reduction balance if my mortgage balance is $ 1 1 7 , 0 0 0 at a mortgage rate of 6 . 2 5 % and I make my payment of 9 3 6

Study smarter with the SolutionInn App


