Show all of the steps in the mechanism for this reaction: CH 3 CH, C-CI+ CHOH CH3
Question:
Show all of the steps in the mechanism for this reaction:
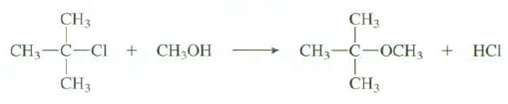
Transcribed Image Text:
CH 3 CH, C-CI+ CH₂OH CH3 CH₂ CH3-C-OCH3 + HCI T CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
CH3 CH3CCH3 Cl CH...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show all of the steps in the mechanism for this reaction and explain the regiochemistry of theaddition: Br Br + HBr
-
Show all of the steps in the mechanism for this reaction, what other products would you expect to beformed? CH3 Br CH3 . CH,O -CH,CH CH.C- C=C CH,CH3 CH3 .
-
(a) Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Who was considered the nemesis of Booker T. Washington in The Awakening of the New Negro and why?
-
Explain how the direct material conversion and total cost per equivalent unit shown in Exhibit 5.3 were calculated.
-
During the current year, Zeta Corporation distributes the assets listed below to its sole shareholder, Susan. For each asset listed, determine the gross income recognized by Susan, her basis in the...
-
The centrifugal pump shown in Fig. P12.18 is not selfpriming. That is, if the water is drained from the pump and pipe as shown in Fig. P12.18(a), the pump will not draw the water into the pump and...
-
Leppard Enterprises had the following selected transactions. 1. Kim Leppard invested $5,000 cash in the business in exchange for common stock. 2. Paid office rent of $950. 3. Performed consulting...
-
Internal auditors are often used to review an organization's financial statements such as balance sheets, income statements, and cash flow statements prior to public filings. Auditors seek to verify...
-
A dc generator has an internal resistance R ohms and develops an open circuit voltage of V volts (Fig. 2.10). Find the value of the load resistance for which the power delivered by the generator...
-
Show the products and the mechanisms of the following reactions. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. CI a) CHCHCHCH SH SN2 OT's c)...
-
Explain why the carbocation shown in Figure 8.8 has a longer lifetime than it does under the conditions shown in Figure 9.8.
-
During its year ended 31 May 2024, Algard plc undertook the following transactions: (i) Issued 50,000 50p ordinary shares at a price of 80p per share. (ii) Redeemed 200,000 5% loan notes at par....
-
A series RLC circuit is driven by an AC source at resonance. The resistor has a resistance of \(R=10 \Omega\), and the AC source has an emf amplitude of \(\mathscr{E}_{\max }=12 \mathrm{~V}\). What...
-
An \(R L C\) circuit consists of a \(20-\mu \mathrm{F}\) capacitor, a \(300-\Omega\) resistor, and a \(50-\mathrm{mH}\) inductor connected in series with an \(A C\) source. What is the resonant...
-
What are heuristics and how might they affect consumer decision-making?
-
An RLC circuit consists of a \(200-\Omega\) resistor, a \(300-\mu \mathrm{F}\) capacitor, and a \(3.00-\mathrm{H}\) inductor. The circuit is connected to a power outlet that has a peak emf of \(170...
-
The resonant angular frequency is \(\omega_{0}\) in identical \(R L C\) circuits 1 and 2 . Because the power delivered by the source at resonance is too high, you reduce the angular frequency of the...
-
Find the indicated critical value. z 0.025
-
Funds are separate fiscal and accounting entities, each with its own self-balancing set of accounts. The newly established Society for Ethical Teachings maintains two funds-a general fund for...
-
What percentage goes to savings? How much money is that? Monthly Budget = $4,000 UTILITIES 10% FOOD 25% CAR 12% SAVINGS MORTGAGE 40%
-
How would you carry out the following transformation? More than one step isneeded. CH3CH2CH2CH2C=CH T ? CCH2CH2CH2 H
-
How would you carry out the following conversions? More than one step is needed in eachcase. "
-
Synthesize the following compounds using 1-butyne as the only source of carbon, along with any inorganic reagents you need. More than one step may be needed. (a) 1, 1, 2, 2-Tetrachlorobutane (b) 1, 1...
-
Salmon ASA has just issued a callable seven-year, 8% coupon bond with coupon payable annually. The bond can be called at par in two years or anytime thereafter on a coupon payment date. It has a...
-
Lamda corporation wants to acquire another company within its industry for $100m and it expects the acquisition to contribute to its free cash flow by $5m the first year, and this contribution is...
-
Dewan INC. has several divisions, each with a manager responsible for the operations of the division. Each division of Dewan controls product design, sales, pricing, operating costs, and profits.....

Study smarter with the SolutionInn App


