Show both the substitution and elimination products that would be formed in these reactions: to , -CI
Question:
Show both the substitution and elimination products that would be formed in these reactions:
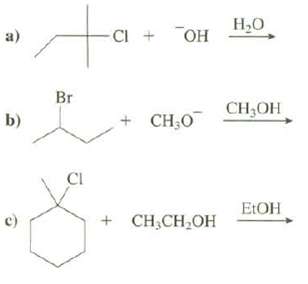
Transcribed Image Text:
to Н,О -CI он a) Br CH,OH CH,O b) CI E:OH + CH;CH,OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
b substituti...View the full answer

Answered By

Simon kingori
I am a tier-one market researcher and content developer who has been in this field for the last six years. I’ve run the freelancing gamut; from market research, data mining and SEO/SMM to copywriting, Content Development, you name it, I’ve done it. I’m extremely motivated, organized and disciplined – you have to be to work from home. My experience in Freelancing is invaluable- but what makes me a cut above the rest is my passion to deliver quality results to all my clients- it’s important to note, I've never had a dissatisfied client. Backed by a Masters degree in Computer Science from MOI university, I have the required skill set and burning passion and desire to deliver the best results for my clients. This is the reason why I am a cut above the rest. Having taken a Bsc. in computer science and statistics, I deal with all round fields in the IT category. It is a field i enjoy working in as it is dynamic and new things present themselves every day for research and exploration.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show both the substitution and elimination products that are formed in these reactions: a) C CI + CH0 CHOH + OH HO EtOH b) Br + CHOH CH3OH
-
Show the aldol addition product that would be formed from each of the following compounds: a. b. c. d. CHCHCH-CHCH - - CH CHCH2CH CH CH3 ,
-
Give the substitution and elimination products for the following reactions, showing the configuration of each product: a. b. c. d. e. f. CH30 (S)-2-chlorohexane /E2conditions CH3OH (S)-2-chlorohexane...
-
The system shown consists of 3 cables. For example; cable C12 joins points 1 and 2. The coordinates of point 1 are (6.4, 0, 0) m, those of point 2 are (0, 9.5, -6.1) m, and those of point 3 are (0,...
-
How does the accounting for spoilage differ under the weighted average and FIFO methods of process costing?
-
Kim retires from the KLM Partnership on January 1 of the current year. At that time, her basis in the partnership is $75,000, which includes her share of liabilities. The partnership reports the...
-
Supersonic airflow enters an adiabatic, constant area pipe (inside diameter \(=0.1 \mathrm{~m}\) ) with \(\mathrm{Ma}_{1}=2.0\). The pipe friction factor is 0.02 . If a standing normal shock is...
-
All jobs at Frankfurt Inc., which uses a job order costing system, go through two departments (Fabrication and Assembly). Overhead is applied to jobs based on machine hours in Fabrication and on...
-
Using the following table, calculate the missing values for Jake's 15year student loan for $13,000.00. The annual interest rate is 7.288%, compounded each month. Find the data missing from the table...
-
Stowe Automotive is considering an offer from Indula to build a plant making automotive parts for use in that country. In preparation for a final decision, Stowes economists have been hard at work...
-
Show all of the steps in the mechanism and explain the stereo chemistry for this reaction: Br , Br + . + C-C CH3 . CH3 .
-
The reaction of 3-iodo-2, 2-dimethylbutane with ethanol gives three elimination products in addition to two substitution products as shown in the following equation. Show all the steps in the...
-
Draw the electron dot formula and the structural formula for a carbon dioxide molecule, CO 2 .
-
How do the PESTEL analysis and Five Forces analysis contribute to the SWOT analysis?
-
What are the differences in the scope of opportunities and threats versus strengths and weaknesses?
-
What is strategic intent? How is it different from a mission statement?
-
How does strategic planning vary at each level of an organization?
-
When should qualitative research be chosen rather than quantitative methods?
-
\(H_{0}: p=0.6\) versus \(H_{1}: p>0.6\) \(n=250 ; x=165\) Test the hypothesis at the \(\alpha=0.05\) level of significance, using (a) the classical approach (b) the P-value approach. Be sure to...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
Sketch the graph of each line. y = 5.x -6- --- L m 2 11 x 9 STE t
-
Give the structures of the possible Claisen condensation products from the following reactions. Tell which, if any, you would expect to predominate in each case. (a) CH3CO2Et + CH3CH2CO2Et (b)...
-
Ethyl dimethylacetoacetate reacts instantly at room temperature when treated with ethoxide ion to yield two products, ethyl acetate and ethyl 2-mcthylpropanoatc. Propose a mechanism for this...
-
In contrast to the rapid reaction shown in Problem 23.40, ethyl acetoacetate requires a temperature over 150 ?C to undergo the same kind of cleavage reaction. How can you explain the difference in...
-
Create a concept map about the topic Continuity of a Function.
-
What role does access control play in operating system security, and how do models like discretionary access control (DAC) and mandatory access control (MAC) differ in their approaches to securing...
-
f(-2) using the given graph

Study smarter with the SolutionInn App


