Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
Question:
Show energy level diagrams for the MOS of these compounds:
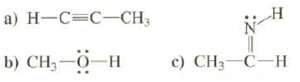
Transcribed Image Text:
a) H-C=C-CH3 b) CH—0—H c) CH3-C-H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a HCCCH3 E 1 c H CH3CH ...View the full answer

Answered By

Allan Simiyu
I am an adroit Writer. I am a dedicated writer having worked as a writer for 3 years now. With this, I am sure to ace in the field by helping students break down abstract concepts into simpler ideas.
5.00+
8+ Reviews
54+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show energy level diagram for the MOs of these compounds? a) H-C=N: b) H 0: C CH3 ) CHNH,
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Draw molecular orbital energy level diagrams for O2, O2-, and O22-.
-
Cindy purchased a building for $1,000,000. How should she deal with the expense on her tax return? a) She must deduct the $1,000,000 in the year of purchase. b) She must not deduct anything and wait...
-
Explain the meaning of the variable overhead efficiency variance?
-
Use the Outsourcing Decision Model Excel file to compute the cost of in house manufacturing and outsourcing for the following levels of demand: 800, 1000, 1200, and 1400. Use this information to set...
-
Water flows in a rectangular channel with a flowrate per unit width of \(q=1.5 \mathrm{~m}^{2} / \mathrm{s}\) and a depth of \(0.5 \mathrm{~m}\) at section (1). The head loss between sections (1) and...
-
Shader Storage Technologies produces refrigeration units for food producers and retail food establishments. The overall average temperature that these units maintain is 46 Fahrenheit. The average...
-
Sale in kg 4 , 0 0 , 0 0 0 Selling price per kg is 3 2 rsFixed cost is 4 , 5 0 , 0 0 0 Variable cost per kg Rs . 2 4 Debt is Rs . 1 0 , 0 0 , 0 0 0 Interest rate is 1 2 % Calculate degree of...
-
Download the rejected loans dataset of LendingClub data titled RejectStatsA Ready from the Connect website and do an Excel PivotTable by state; then figure out the number of rejected applications for...
-
Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
The Sanchez Computer Center currently has an $11,900 balance in Accounts Receivable. Here is a current schedule of Accounts Receivable: Sanchez Computer Center Schedule of Accounts Receivable March...
-
Engineers are often accused of having a narrow view of problems, and are claimed to lack skills to deal with ambiguity. In what ways might an engineering manager differ in this regard?
-
It is mentioned that for a "decision to be made" there must be more than one alternative under consideration and the possible outcomes must be of unequal value. What are the three general types of...
-
How might the market value of a firm differ from its intrinsic value?
-
It was stated that the topic of tax accounting would not receive extensive treatment in this textbook. In what way could this be justified? What might be the impact on what can be learned from the...
-
Intuition in decision-making can expedite decision-making (versus cognitive domains that are slow and deliberate), but it can fail or mislead us, too. How can that be?
-
Two-tailed test with \(\alpha=0.05\), degrees of freedom in the numerator \(=12\), degrees of freedom in the denominator \(=20\). Find the critical value( \(s\) ) for \(\alpha\).
-
Using Gauss-Jordan elimination, invert this matrix ONLY 0 0 0 0 1
-
Find the sum of the series correct to three decimal places. (By definition, 0! = 1.) 00 n=0 (-1)" n!
-
Names the following substances, including the cis-or trans-prefix (red-brown =Br): (b) (a)
-
Each H H eclipsing interaction in ethane costs about 4.0kJ/mol. How many such interactions are present in cyclopropane? What fraction of the overall 115 kJ/mol (27.5kcal/mol) strain energy of...
-
Cis-l, 2-Dimethylcyclopropane has more strain than trans-1, 2-dimethylcyclo- propane. How can you account for this difference? Which of the two compounds is more stable?
-
Supply is the connection between the cost of a decent and the amount ready to move costing that much. It very well might be addressed as a table or diagram relating cost and amount provided. Makers,...
-
If 50% of the men 18-34 years old watch television on Mondays at 11 PM, and 30% of those viewers watch Monday Night Football, what is Monday Night Football's rating among men 18-34?
-
Assume you are a media planner and you have just completed a media plan using both magazine and television advertising. Your magazine schedule reaches 50 percent of the target audience and your...

Study smarter with the SolutionInn App


