Explain why one of these carbocations is much more stable than the other: + CH-CH3 CH CH
Question:
Explain why one of these carbocations is much more stable than the other:
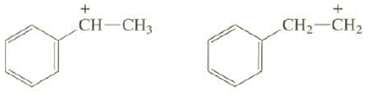
Transcribed Image Text:
+ CH-CH3 CH₂ CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
The p orbital of the positively charged carbon of the carbocation on the left is con...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why one of these anions is much more stable than the other: : a) CH-C-CH-CH b) CH CH3 : CH-C-CH-CH CH-C=N:
-
One of the following compounds is much more stable than the other two. Classify each as aromatic, antiaromatic, or nonaromatic. heptalene azulene pentalene
-
Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide in ethanol but the other doesnot: OTs OTS . H, .H, CH; CH3
-
Amrito Corporation is under financial distress and raises debt because it has several projects that are expected to generate profit in the future. When calculating its weighted average cost of...
-
Define the fixed overhead budget variance, and explain how it may assist manager control costs?
-
For the Shafer Office Supplies, suppose that the company wants to enforce a single sourcing constraint that each retail store be served only from one distribution center. Set up and solve a model to...
-
Water flows in a horizontal rectangular channel with a flowrate per unit width of \(q=10 \mathrm{ft}^{2} / \mathrm{s}\) and a depth of \(1.0 \mathrm{ft}\) at the downstream section (2). The head loss...
-
At the beginning of 2011, the Healthy Life Food Company purchased equipment for $42 million to be used in the manufacture of a new line of gourmet frozen foods. The equipment was estimated to have a...
-
Did the government have an easy time convicting Elizabeth Holmes?
-
A company has three items in its inventory that are purchased from the same vendor and are shipped together on the same delivery truck. The truck has a capacity of 30,000 lb. The items are under the...
-
Explain why this carbocation is considerably more stable than this structure would suggest: H +C-0-CH, H
-
Show energy level diagrams for the MOS of these compounds: a) H-C=C-CH3 b) CH0H c) CH3-C-H
-
Construct a preference table with three candidates and four rankings of the candidates, similar to the preference table in Exercise 10, such that the plurality with elimination method violates the...
-
How would a company know that its product or service was preferred in the marketplace? What could a competitor do to erode this favorable position?
-
It is claimed that problem definition and decision-making are intertwined. Explain.
-
Clarify the difference between risk and regret.
-
Why are most engineers likely to have experience with deterministic risk and not probabilistic risk?
-
Managers must be problem-solvers but are not always decision-makers. Do you agree? Disagree? Provide an example that clarifies your position.
-
Right-tailed test with \(\alpha=0.025\), degrees of freedom in the numerator \(=15\), degrees of freedom in the denominator \(=50\). Find the critical value( \(s\) ) for \(\alpha\).
-
Cable Corporation is 60% owned by Anna and 40% owned by Jim, who are unrelated. It has noncash assets, which it sells to an unrelated purchaser for $100,000 in cash and $900,000 in installment...
-
Find the sum of the geometric series 5 10/3 + 20/9 40/27 + .....
-
Name the following substances, including the cis- or trans-prefix: CH2CH3 (b) , (a) H. - - CI
-
Draw the structures of the following molecules: (a) Trans-1-Bromo-3-methylcyclohexane (b) Cis-1, 2-Dirnethylcyclobutane (c) Trails-1-tert-Butyl-2-ethylcyclohexane
-
Prostaglandin F2?, a hormone that causes uterine contraction during childbirth, has the following structure. Are the two hydroxyl groups (?OH) on the cyclopentane ring cis or trans to each other?...
-
Scenario: Yarning Circle and Talking Stick Sarah has recently been employed by Blue Bay Early Learning Centre. Sarah is very proud of having just achieved the Diploma of Children's Services and she...
-
The principal at your school decides to retire, and several well-qualified teachers on the staff apply for the position. Mr. England has been teaching for four years and has his administrator's...
-
Sandi's parents have been divorced since she was five years old. She is now seven. She lives with her mother and visits her father every other weekend. He brings her to school on Monday mornings...

Study smarter with the SolutionInn App


