Show how these compounds could be synthesized from alkylhalides: a) Heptane b) PHCH,CH,CH,NH, c) PHCH,C=CH d) e)
Question:
Show how these compounds could be synthesized from alkylhalides:
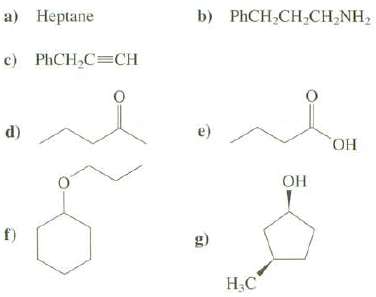
Transcribed Image Text:
a) Heptane b) PHCH,CH,CH,NH, c) PHCH,C=CH d) e) HO, OH f) g) H.C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
a b PhCHCHCHBr c PhCHCl Br 1 LIAH 2 HO ...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how the following compounds could be synthesized from phenylacetylene (C6H5CCH): (a) 1-phenylpropyne, (b) 1-phenyl-1-butyne, (c) (Z )-1-phenylpropene, and (d) (E )-1-phenylpropene. Begin each...
-
Show how the following compounds could be synthesized from benzene: a. m-dibromobenzene b. m-bromophenol c. o-chlorophenol d. m-nitrotoluene e. p-methylbenzonitrile f. m-chlorobenzaldehyde
-
Show how the following compounds could be synthesized from benzene: a. m-chlorobenzenesulfonic acid b. m-chloroethylbenzene c. benzyl alcohol d. m-bromobenzonitrile e. 1-phenylpentane f....
-
A bond currently has a price of $940. The current yield to maturity on the bond is 7%. If the yield decreases by 20 basis points, the price of the bond will go up to $960. Based on this information,...
-
Madigan Company commenced operations at the beginning of the current year. One of the company's products, a refrigeration element, sells for $185 per unit, Information related to the current year's...
-
A manufacturer claims that the mean operating cost per mile of its minivans is less than that of its leading competitor. You conduct a study using 34 randomly selected minivans from the manufacturer...
-
What are the benefits of using value management in projects?
-
Barbara is considering investing in a stock and is aware that the return on that investment is particularly sensitive to how the economy is performing. Her analysis suggests that four states of the...
-
q security analyst reviews a firewall log's source IP addresses to investigate an attack. These logs are a representation of what type of functional security control?
-
The following balance sheet, which has some weaknesses in terminology and classification, has been prepared by an inexperienced accountant and submitted to you for review: Required: Prepare a...
-
Show reactions that could be used to convert the epoxide to these compounds. More than one step may benecessary. SCH3 OH b) CH,O- a) Br
-
Show how this synthesis might beaccomplished: Br CH3 CH3 HO from "CN -
-
Light with a wavelength of 405 nm fell on a strontium surface, and electrons were ejected. If the speed of an ejected electron is 3.36 105 m/s, what energy was expended in removing the electron from...
-
DATE 1 April 5 April 10 April 15 April 20 April 25 April 30 April TOTAL AMOUNT 12.000,00 5.000,00 15.000,00 INCOMING UNIT COST 25,00 26,00 22,20 TOTAL AMOUNT 10.000,00 12.000,00 8.000,00 OUTGOING...
-
At September 30, 2023, Fern Field Farms, Inc. reported $142,000 of cash, $78,000 of other assets, $120,000 of liabilities, and $100,000 of common stock. Prepare the company's September 30, 2023...
-
1.9- Martin purchased a van for 12,000 during his six-month period of account to 31 July 2023. What is the maximum amount of capital allowances available for the van for the six months ended 31 July...
-
1.3 - Andi Ltd manufactures hair products, which are standard-rated for VAT. The products are sold to Bart Ltd, a wholesaler. Charlotte purchases these goods to use in her hairdressing business,...
-
During the morning peak hour, a total of 5 , 5 0 0 person - trips are made to downtown along a major arterial in the city: 4 5 0 0 person - trips are made by car and 1 0 0 0 person - trips are made...
-
Describe the nature of interrogatories and identify their strengths and weaknesses.
-
a. Why does the Wi-Fi Alliance release compatibility testing profiles in waves instead of combining the entire standards features initially? 27a1.) An 802.11ac Wi-Fi compatibility testing profile...
-
Solve each equation. -6(6b-8)= -3b + 3(2b + 3)
-
A Propose a structure for an alkyl halide that gives only (E)-3-methyl-2-phenyl-2-pentene on E2 elimination, Make sure you indicate the stereochemistry.
-
One step in the urea cycle for ridding the body of ammonia is the conversion of arginino succinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the...
-
Although anti periplanar geometry is preferred for E2 reactions, it isn?t absolutely necessary. The deuterated bromo compound shown here reacts with strong base to yield an un-deuterated alkene....
-
Evaluate economic conditions that influence company performance of starbucks Consider political, environmental, currency ( money ) , global economics, and government influences on economic...
-
(Part 1) : The business letter to your employer. The purpose of the letter is to explain why diversity is important in the workplace and to recommend actions the organization can take to become more...
-
Question 4 (20 marks)In the recent annual general meeting of Umi Technology Limited, the board of directors declared an annual dividend of $5.00 per share, and the shareholders agreed to a rights...

Study smarter with the SolutionInn App


