Although anti periplanar geometry is preferred for E2 reactions, it isn?t absolutely necessary. The deuterated bromo compound
Question:
Although anti periplanar geometry is preferred for E2 reactions, it isn?t absolutely necessary. The deuterated bromo compound shown here reacts with strong base to yield an un-deuterated alkene. Clearly, a syn elimination has occurred. Make a molecular model of the reactant, and explain the result.
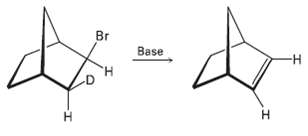
Transcribed Image Text:
Br Base -н Н н т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (14 reviews)
One of the steric requirements of E2 elimination is the need ...View the full answer

Answered By

Ashish Jaiswal
I have completed B.Sc in mathematics and Master in Computer Science.
4.90+
20+ Reviews
39+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The triene shown here reacts with 2 equivalents of maleic an hydride to yield a product with the formula C17H16O6. Predict a structure for theproduct. CH2 C17H1606 C=CH2 2. U
-
Give the structure or make a molecular model of the most stable C6H12 alkene.
-
A compound with molecular formula C4H8O has a strong IR absorption at 1730 cm-1. Its mass spectrum includes key peaks at m/z 44 (the base peak) and m/z 29. Propose a structure for the compound and...
-
2.) A truck with axle loads of W = 19.6kN and W2 = 78.6kN on a wheel base of d = 4.3m rolls across the beam shown in the figure. Determine the following: 3m 25 m 22 m a.) Draw the influence line for...
-
What were the training objectives for salespeople? How did ConAgra measure the results of training them?
-
Explain the following observations: When tert-butyl bromide is treated with sodium methoxide in a mixture of methanol and water, the rate of formation of tert-butyl alcohol and tert-butyl methyl...
-
Which of the following accounting issues is most likely to raise an auditor's professional skepticism about earnings manipulation? a. Progress payments. b. Allowance for doubtful accounts. c. Sales...
-
Journalizing Various Receivable Transactions Presented below is information related to Sanford Corp. July 1 Sanford Corp. sold to Legler Co. merchandise having a sales price of $10,000 with terms...
-
The balance of owner's capital at the beginning of the year was $50,000 and at the end of the year was $67,000. No additional capital was contributed during the year. Withdrawals were $23,000. What...
-
Name three applications for feedback control systems.
-
One step in the urea cycle for ridding the body of ammonia is the conversion of arginino succinate to the amino acid arginine plus fumarate. Propose a mechanism for the reaction, and show the...
-
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy? (a) CI...
-
Suppose we introduce one carbon atom for every 100 iron atoms in an interstitial position in BCC iron, giving a lattice parameter of 0.2867 nm. For the Fe-C alloy, find the density and the packing...
-
A pendulum is oscillating with its length 120 cm on Earth. (a) Find out its time period. (b) If pendulum is taken to Mars (g = 3.7 m/s^2), what would be its time period? 10. Ellie is pushing a...
-
What rhetorical techniques can be employed in business writing to effectively persuade stakeholders? Discuss how these techniques differ when addressing internal versus external audiences.
-
A Ford passes a Toyota on the road (both vehicles are traveling in the same direction). The Ford moves at a constant speed of 47.4 m/s. Just as the Ford passes it, the Toyota is traveling at 18.5...
-
Driving down the road at a speed of 30.1 m/s, you suddenly notice a fallen tree blocking the road a distance of 55.1 m ahead of you. You step on the brake pedal and decelerate at a constant rate....
-
On planet #2, you launch a projectile straight up from the ground at a speed of 43.3 m/s. The projectile reaches a maximum height of 63.3 m before falling back to the ground. What is the value of g...
-
A spaceship flies past an experimenter who measures its length to be one-half the length he had measured when the spaceship was at rest. An astronaut aboard the spaceship notes that his clock ticks...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
The following reaction is endothermic: What is the effect of increasing the temperature of the reaction mixture? Decreasing the temperature? CaCO3(s) = CaO(s) + COz(g)
-
Show how the following products might be synthesized from suitable Michael donors and acceptors. (a) (b) (c) (d) (e) (f) Ph CH CH, C OCH,CH CH COOCH,CH CH CH, CN CH COCH, CH,CH CN CH3 CH2CH2 C Ph CH...
-
Propose a mechanism for the following reaction. CH o H,CCH CH,CH,
-
The base-catalyzed reaction of an aldehyde (having no hydrogens) with an anhydride is called the Perkin condensation. Propose a mechanism for the following example of the Perkin condensation. (Sodium...
-
Describe the impact of Non-Uniform Memory Access (NUMA) on an operating system's memory management strategies, and how an OS can optimize performance in a NUMA-based hardware environment .
-
Explain the concept of system calls in operating systems, and discuss the security implications of exposing system-level operations to user space.
-
Rafner Manufacturing has the following budgeted data for its two production departments. Budgeted Data Assembly Overhead cost $ 1,560,000 $ 1,035,000 Direct labor hours Machine hours 13,000 direct...

Study smarter with the SolutionInn App


