Show that g(E) = (3N / 2)EF -3/2 E1/2 (Equation 27-30) follows from Equation 27-28 for g(E),
Question:
Show that g(E) = (3N / 2)EF -3/2 E1/2 (Equation 27-30) follows from Equation 27-28 for g(E), and Equation 27-15a forEF.
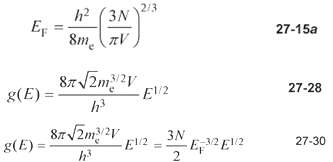
Transcribed Image Text:
h? (3N)2/3 EF 8m (zV %3D 27-15a 87 2m2V g(E) = 27-28 87V2m2V 3N 8(E): 27-30 hi
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 47% (17 reviews)
From Equation 2715 a Subst...View the full answer

Answered By

Rajnish Verma
I have 2.5 years of teaching experience in a coaching institute. Here, I taught students maths up to graduation level. Here I learned about students mindset and difficulties they face while they attend the class. So, I tried to set a comfort level with them.
I have also worked as a maths content developer.
Here, I provide step by step solution to the students questions.
I became familiar with Bloom taxonomy , Math type and judging the difficulty level of the questions.
I got a global exposure and professionalism.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
Show that equation (15.4) follows from equation (15.3) and the initial condition T(0) = 1. (15.4) (1
-
Show that 2n is O(3n) but that 3n is not O(2n). (Note that this is a special case of Exercise 60.)
-
Water at 26C flows in the 3-mm-diameter pipe of Fig. below. The pressure drop over the 15-m section is 6 kPa. Find the Reynolds number of the flow and the wall shear stress. Assume lami - nar flow....
-
Use the information in Figure to answer the following questions: a. What is the six-month forward rate for the Japanese yen in yen per U.S. dollar? Is the yen selling at a premium or a discount?...
-
The Appliance Store began operations March 1, 2019. The firm sells its merchandise for cash and on open account. Sales are subject to a 6 percent sales tax. During March, The Appliance Store engaged...
-
You have the following solutions, all of the same molar concentration: KBr, HBr, CH3NH2, and NH4Cl. Rank them from the lowest to the highest hydroxide-ion concentration.
-
Locate sample affidavits used in a civil litigation for both California court and federal court. What do they have in common? Are there any striking differences?
-
Orasco Company uses both standards and budgets. For the year, estimated production of Product X is 500,000 units. Total estimated cost for materials and labor are $1,200,000 and $1,600,000. Compute...
-
Kraft Heinz was compelled to restate nearly three years of financial reporting following an examination. As a financial analyst, do you believe that restatements of this nature should raise warning...
-
Use the frequency distribution from Exercise 12 in Section 2-1 on page 49 to construct a histogram. Does the histogram appear to be skewed? If so, identify the type of skewness. In construct the...
-
What is the difference between the energies at which the Fermi factor is 0.9 and 0.1 at 300 K in (a) Copper, (b) Potassium, and (c) Aluminum.
-
The density of the electron states in a metal can be written g(E) = AE1/2, where A is a constant and E is measured from the bottom of the conduction band. (a) Show that the total number of states is...
-
Suppose the life of a particular brand of calculator battery is approximately normally distributed with a mean of 75 hours and a standard deviation of 10 hours. a. What is the probability that a...
-
A $1,000 par value bond has an 8% coupon rate (paid annually). It has 10 years remaining to maturity. If the bond's current price is $935.82, what should be the YTM of this bond?
-
1. The melting point of pure trans-cinnamic acid is 133-134C and that of 2-acetoxybenzoic acid is 134-136C. Describe a melting point procedure by which you could identify whether an unknown sample...
-
CBI-3 In most metals there are one or two conduction (free) electrons per atom with a total of something like 1026 free electrons in a 10 gram piece of the metal. These electrons are free to move...
-
how long, in years, will it take an initial depoit of % 5 0 0 0 into an account that earns 4 . 7 5 % compounded monthly to grow to $ 1 0 , 0 0 0 , if no other funds are deposited, and no withdrawals...
-
Sit Down Publishers publishes The Accountant magazine and sells one - year ddsubscriptions to its customers. Customers are required to pay in full for the one - year subscription at the beginning of...
-
A plastic spring with spring constant \(k=450 \mathrm{~N} / \mathrm{m}\) has a relaxed length of \(0.100 \mathrm{~m}\). The spring is positioned vertically on a table, and a charged plastic...
-
What is the difference between adsorption and absorption?
-
Carry out the following algebraic manipulations. Given p/q = r, solve for p.
-
Describe all the forces acting on a child riding a horse on a merry-go-round. Which of these forces provides the centripetal acceleration of the child?
-
A bucket of water can be whirled in a vertical circle without the water spilling out, even at the top of the circle when the bucket is upside down. Explain.
-
How many accelerators do you have in your car? There are at least three controls in the car which can be used to cause the car to accelerate. What are they? What accelerations do they produce?
-
1. What is the importance of keeping in mind the risks associated with collaborations? 2. Why is it crucial to assess the strategic importance of existing relationships? 3. What should an...
-
When would it be better to have a checking account with no per-check fee and a monthly fee, rather than a checking account that is cost-per-check?
-
What are 3 ways to engage the community in assessing a project after it is completed? What project issues do you believe have the biggest effect on time and cost? Why? What are some ways to avoid...

Study smarter with the SolutionInn App


