Show the products, including stereo chemistry, of these SN2 reactions: a) b) c) HC Ph CH H
Question:
Show the products, including stereo chemistry, of these SN2 reactions:
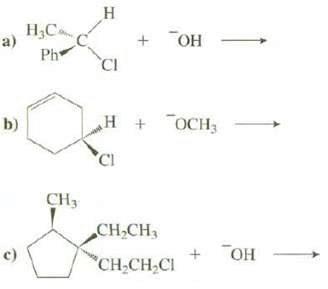
Transcribed Image Text:
a) b) c) H₂C Ph CH₂ H CI + OH 2H + OCH3 н CH₂CH₂ CH₂CH₂Cl ТОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
SN2 reactions always occur with inversion of configuration at the reaction center The nucleophile ap...View the full answer

Answered By

Wonder Dzidzormenu
As a professional accountant and a teacher, I explain account ing concepts in a more practical way that makes students more connected to the subject.
With over 10 years of teaching accounting , I offer a well constructed , easily understood and in-depth explanations to students questions.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show how you might use SN2 reactions to convert 1-chlorobutane into the following compounds. (a) Butan-1-ol (b) 1-fluorobutane (c) 1-iodobutane (d) CH3-(CH2)3-CN (e) (f) CH3CH2-O-(CH2)3-CH3 (g) CH3-...
-
SN2 reactions that involve breaking a bond to a chirality center can be used to relate configurations of molecules because the stereochemistry of the reaction is known. (a) Illustrate how this is...
-
In contrast to SN2 reactions, SN1 reactions show relatively little nucleophile selectivity. That is, when more than one nucleophile is present in the reaction medium, SN1 reactions show only a slight...
-
(a) Using your knowledge of economics and how markets work illustrate and explain why the price of electricity has increased so much over the past 18 months throughout the EU. You should consider the...
-
'Despite the use of predetermined overhead rates in estimating service costs, there is no need to consider underapplied or overapplied overhead.' Do you agree with this statement? Explain your...
-
Dene a deferred expense. Provide two examples of deferred expenses. Why are adjusting entries generally required at the end of an accounting period for deferred expenses?
-
Compare Colburn Eq. (16-81d) to the equivalent Kremser Eq. (13-9c), and compare Eq. (16-81e) to \((13-9 b)\). If we relate \(\mathrm{n}_{\mathrm{O}-\mathrm{Ey}}\) to \(\mathrm{N}\) and set...
-
Prepare adjusting journal entries for the year ended (date of) December 31, 2013, for each of these separate situations. Assume that prepaid expenses are initially recorded in asset accounts. Also...
-
Help me solve this step by step please and thank you! At January 1, 2024, Page Inc. reported the following information on its statement of financial position: Accounts receivable $520,000 Allowance...
-
The wing of a small plane is represented by a simplified prismatic cantilever beam model acted on by the distributed loads shown in the figure. Assume constant EI = 1200 kN*m 2 . Find the tip...
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
Explain which compound has a faster rate of SN2 reaction: a) HC CI CI c) PhCHCH3 or H CI Cl or CHCHCH3 b) CI CH3 CH3 or U
-
Pandora Corporation operates several factories in the Midwest that manufacture consumer electronics. The December 31, 2016, year-end trial balance contained the following income statement items:...
-
2C distributes laptops to retail stores and extends credit terms 3/15, n/60 to all its customers. During the month of June 2021, the following merchandising transactions occurred: June 12: Purchased...
-
Courtney has decided that she wishes to travel to Malaysia for 4 months when she finishes university. Courtney has estimated that her trip expenses will be $1500.00 per month of travel, and she will...
-
3. What are the potential pitfalls for the underwriter of all the information made available by technology
-
(30) Nelson Corporation sells three different products. The following information is available on December 31: Inventory Item Units X Y Z Cost per unit Net realizable value per unit 300 $4.00 $3.00...
-
what is the fair price of te futures contract based on the s&p500 which expires in 3 months time
-
The mass matrix of a vibrating system is given by \[[m]=\left[\begin{array}{lll}1 & 0 & 0 \\0 & 2 & 0 \\0 & 0 & 1\end{array} ight]\] and the eigenvectors by Find the \([m]\)-orthonormal modal matrix...
-
Danielle has an insurance policy with a premium of $75 per month. In September she is in an accident and receives a bill worth $2990 for the repair of her own property. Her deductible is $250 and her...
-
Solve each equation. 5 2r+- 4 65 8
-
Draw both cis- and trans-1, 3-dimethylcyclohexane in their most stable chair conformations. (a) How many stereo isomers are there of cis-l, 3-dimethylcyclohcxane, and how many of trans-1,...
-
Cis-1, 2-Dimethylcyclohexane is optically inactive even though it has two chirality centers. Explain.
-
Well see in the next chapter that alkyl halides react with nucleophiles to give substitution products by a mechanism that involves inversion of stereochemistry at carbon: Draw the reaction of...
-
13. A present investment of $50,000 is expected to yield $7,000 per year at the end of each year for 15 years. Determine the annual rate of return. 6500
-
5 20 points Suppose that you are a U.S.-based importer of goods from the United Kingdom. You expect the value of the pound to increase against the U.S. dollar over the next 30 days. You will be...
-
Problem 7-06 As an equity analyst, you have developed the following return forecasts and risk estimates for two different stock mutual funds (Fund T and Fund U): Forecasted Return Fund T Fund U 7.0%...

Study smarter with the SolutionInn App


