Show the products of thesereactions: H,SO,, H,SO4 b) + H,0 + H,0 H,SO. + HO
Question:
Show the products of thesereactions:
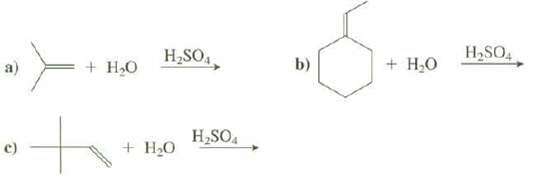
Transcribed Image Text:
H,SO,, H,SO4 b) + H,0 + H,0 H,SO. + HO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
These are acid catalyzed hydrolysis reactions where th...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Assume a retailer has fixed costs of $10,000, a unitvariable cost of $25, and a 50% retail margin. How many units must be sold for her tobreak-even? If she has a target profit of $200,000, how many...
-
Faced with rising pressure for a $15 per hour minimum wage rate, the farming industry is currently exploring the possible use of robotics to replace some farm workers. The Lettuce Bot is one such...
-
Why do people give gifts?
-
When is validation started and when does it end?
-
Performance Measures. Information from the Form 990 for the American Heart Association for the fiscal year ending June 30, 2007, follows. Required a. Compute the following performance measures using...
-
4. A lumber has a nominal dimension of 2x4 inches and is tested for 3-point bending with a span of 4 ft. The wood does not get ruptured until the point load reaches 88 lbs. (Note: The 2-inch side of...
-
Consider the following 0x86 program: .data array DWORD 1,2,3,4,5,6,7,8,9 aravSize -(S-array)/4 ; array .code main PROC mov ecx,arraySize-1 mov esi.OFFSET array L1: mov eax. [esi] sda mov bx,2 idiv bx...
-
Show the products of thesereactions: CH3 HBr a) CH,CHCH-CH2 HBr b) PhC CH 2 HBr
-
Show all of these steps in the mechanism for the addition of water to propene catalyzed by sulfuric acid. Explain whether propene or phenylethene (PhCH = CH2) has a faster rate in this reaction:
-
A floor truss is loaded as shown. Determine the force in members FI, HI, and HJ. I EN 2 kN 2 kN 15KN I KN I kN 0.5 kN Im Im Im Im1 m Im 0.5
-
Kimo is a parent who has $200 in non-labor income. Kimo can earn $10 per hour working between 0 and 60 hours in a week if he works. Draw and label clearly the potential income constraint diagram...
-
The next step traditional airlines take to regain the lost market is to invent cheaper class fares. You can still get all the "extra" services for free on most traditional airlines today, such as...
-
Assume the price of belt buckles falls to S4 and the minimum wage remains $10. Complete the table using the given information. How many workers should you hire to maximize profit?
-
Central banks and governments can have a large influence on market movements. Financial advisors should understand the main types of policies and how these can impact markets. What are the most...
-
In Exhibit 13-4, the makers of Healthy Hands Lotion discovered that the lotion can cause skin reactions, but it doesn't inform the buyers. While the market experiences imperfect information, the...
-
At room temperature, do collisions between nitrogen molecules have enough energy to set a nitrogen molecule rotating? Enough to set it vibrating? The quantum of energy associated with rotation is...
-
The Adjusted Trial Balance columns of a 10-column work sheet for Webber Co. follow. Complete the work sheet by extending the account balances into the appropriate financial statement columns and by...
-
Calculate the molar solubility of Co(OH) 3 , K sp = 2.5 10 -43 .
-
How would you prepare the following compounds from benzene, using a diazonium replacement reaction in your scheme? (a) p-Bromobenzoic acid (b) m-Bromobenzoic acid (c) n-Bromo chloro benzene (d)...
-
Propose a synthesis of p-(dim-ethylamine) azobenzene from benzene as your only organic starting material.
-
Draw an orbital picture of thiazole. Assume that both the nitrogen and sulfur atoms are sp2-hyhridized, and show the orbitals that the lone pairs occupy.
-
A large hospital required nurses to work 80 hours over two-week (14 day) pay periods. In addition, nurses were also considered to be "on-call" for an additional 20 hours during the pay period. While...
-
Describe the history of the HIPAA, explaining why it is essential. Consider the following questions in your response: Why is understanding the history of the HIPAA law essential in decision making...
-
"I'm not sure we should lay out $265,000 for that automated welding machine," said Jim Alder, president of the Superior Equipment Company. "That's a lot of money, and it would cost us $75,000 for...

Study smarter with the SolutionInn App


