Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) CI Br ELOH + NaOCH,CH; b)
Question:
Show the products of these eliminationreactions:
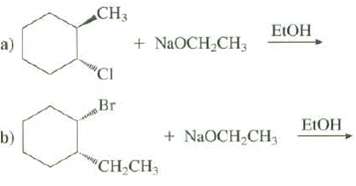
Transcribed Image Text:
CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
For anti elimination to occur in a cyclohexane ring the hyd...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these elimination reactions and indicate which ismajor: OTs . b) CH,OH + CH,0 + OH ELOH CI E:OH + CH,CH,O c)
-
Suppose that a 20-year bond pays $4 coupon once a year, and its yield rate is 6%. The bond has write-down of $0.49 for the first year. Calculate the write-down amount for the third year. Round the...
-
Stingray Ltd manufactures outboard motors and an assortment of other marine equipment. The company uses a job costing system and manufacturing overhead is applied on the basis of machine hours....
-
Carl Carson, a single taxpayer, owns 100% of Delta Corporation. During 2013, Delta reports $150,000 of taxable income. Carl reports no income other than that earned from Delta, and Carl claims the...
-
Briefly explain the main difference between exchange-traded funds (ETFs) and mutual funds?
-
The graph of the probability distribution of a normally distributed random variable with a mean of 20 and standard deviation of 3 is shown in Figure. The Excel function = NORMINV(Rand( ),20,3) also...
-
Production data: Kilograms in process, May 1 (materials 100% complete; conversion 80% complete) Kilograms started into production during May Kilograms completed and transferred to Coating Kilograms...
-
The uniform beam has a mass of 50kg per meter of length. Determine the reactions at the supports. A| -2.4 m- Problem 3/4 1.2 m 300 kg B
-
Both cis and trans alkenes can be formed from this compound y anti elimination. Draw a Newman projection of the conformation required to form each of these products and, on the basis of these...
-
Explain why one of these compounds reacts readily by an E2 mechanism when treated with sodium ethoxide in ethanol but the other doesnot: OTs OTS . H, .H, CH; CH3
-
If one of the students takes the risky job, how much more would the fruit-picking job have needed to pay to attract that student? Two students, Jim and Kim, are offered summer jobs managing a student...
-
Imagine a system that consists of only a single positively charged object. Is it possible to have in this system a closed surface that has a negative field line flux through it? (The charged object...
-
Suppose a certain planar surface has 2000 field lines passing through each square meter, normal to the surface. How many field lines pass through each square meter if the surface is tilted by...
-
Two parallel conducting plates carry equal and opposite charges. The plates are large relative to their separation distance, so we can assume the electric field between them is uniform. The potential...
-
A small charged ball is suspended at the center of a spherical balloon that is nestled snugly inside a cubical cardboard box. On one side, the balloon touches the wall of the box. (a) At this point...
-
Suppose a charged particle is located at the origin. What is the direction of the electric field at the point \((0.6,1.2)\) if the particle is \((a)\) positively charged and \((b)\) negatively...
-
A feed of \(13,500 \mathrm{~kg} / \mathrm{h}\) consists of \(8 \mathrm{wt} \%\) acetic acid in water. Acetic acid will be removed from the solution by extraction with pure methyl isobutyl ketone at...
-
If the joint cost function for two products is C(x, y) = xy2 + 1 dollars (a) Find the marginal cost (function) with respect to x. (b) Find the marginal cost with respect to y.
-
Find the mode, median, mean, lower quartile, upper quartile, and interquartile range for eacl data set. 2 4 2 2 # Words in Book Titles 3 4 2 2 4 6 2 2 2 3 3
-
Two infrared spectra are shown. One is the spectrum of cyclohexane, and the other is the spectrum of cyclohexane. Identify them, and explain youranswer. (a) 100 80 60 20 - 1000 4000 3500 3000 2500...
-
At what approximate positions might the following compounds show IRabsorptions? c??H? " alt="At what approximate positions might the following compounds show IRabsorptions? 51524" class="fr-fic...
-
How would you use infrared spectroscopy to distinguish between the following pairs of constitutionalisomers? (a) CH3C=CCH3 and CHCH2C3CH () CHH CHCCH2HH2 and (c) H CH3CH2CHO and
-
A production department reports the following conversion costs. Equivalent units of production for conversion total 436,000 units this period. Calculate the cost per equivalent unit of production for...
-
If you were asked whether a large university such as Tennessee or Michigan with a large seating capacity for their football stadiums should build a new football stadium, how would you respond and...
-
J is going to receive a 30-year annuity of 8,500 and L is going to receive perpetuity of 8,500. If the appropriate interest rate is 6%, how much more is L's cash flow worth?

Study smarter with the SolutionInn App


