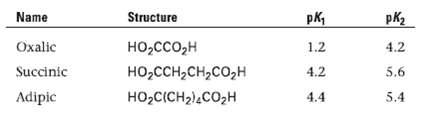
Shown here are some pKa data for simple dibasic acids. How can you account for the fact
Question:
Shown here are some pKa data for simple dibasic acids. How can you account for the fact that the difference between the first and second ionization constants decreases with increasing distance between the carboxyl groups?
Transcribed Image Text:
Name Structure pk, pK2 Oxalic Succinic 4.2 1.2 HO,cco,H но -ссн-сн,со,н но -СICH2),cо,H 4.2 5.6 4.4 Adipic 5.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (14 reviews)
Inductive effects of functional groups are transmitted through bonds For oxalic acid the e...View the full answer

Answered By

SUMAN DINDA
I LIKE TO TEACH STUDENTS. SO, I START MYSELF AS A PRIVATE TUTOR. I TEACH STUDENTS OF DIFFERENT CLASSES. I HAVE ALSO DONE BACHELOR OF EDUCATION DEGREE(B.ED). DURING THIS COURSE I HAD TO TEACH IN A SCHOOL. SO I HAVE A GOOD EXPERIENCE IN TEACHING.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How can you account for the fact that 2, 2, 6-trimethylcyclohexanone yields no detectable aldol product even though it has an acidic hydrogen?
-
How can you account for the fact that cis-1, 3-pentadicne is much less reactive than trans-1, 3-pentadiene in the DielsAlder reaction?
-
How can you account for the fact that normal rain is slightly acidic?
-
The following data applies to the two unrelated companies Lloyd Ltd and Cole Ltd: All taxable and deductible temporary differences relate to the profit or loss. Assume a corporate tax rate of 30%. A....
-
1. To what degree do you agree or disagree with the B-Team's goal for world governments to reach net zero CO2 or greenhouse gas (GHG) emissions by 2050? 2. Should governments of developing economies...
-
Describe each of the following correlation coefficients using the terms strong, moderate, or weak and positive or negative. a. r = 0.17 b. r = - 0.62 c. r = - 0.88 d. r = 0.33 e. r = 0.49 f. r = -...
-
What are the key System Elements of an SOIs Mission System?
-
Identify the primary audit objectives that auditors hope to accomplish by confirming a clients year-end accounts receivable. Explain the difference between positive and negative confirmation requests...
-
Sasha got fired from her sweet job as an accountant, and is now earning only $25 before taxes. If she can work up to 8 hours per day, 5 days per week, draw out the weekly budget constraint on the...
-
Chittenden County had the following federal award activity during the most recent fiscal year: Required a. Based on size, which programs would be considered Type A programs? Type B programs? b. You...
-
In humans, the final product of purine degradation from DNA is uric acid, pKa = 5.61, which is excreted in the urine. What is the percent dissociation of uric acid in urine at a typical pH = 6.0? Why...
-
Predict the product of the reaction of p-methyl benzoic acid with each of the following: (a) LiAlH 4 , then H 3 O + (b) N-Bromosuccinimide in CCl 4 (c) CH 3 MgBr in ether, then H 3 O + (d) KMnO 4 , H...
-
Quinn Becker is president of Better Books. She has no accounting background. Becker cannot understand why fair value is not used as the basis for all accounting measurement and reporting. Discuss.
-
Can you elucidate the symbiotic relationship between autonomy and collaboration in innovative projects?
-
Why does it later rise? How would you describe that in economic terms again?
-
Paul corporation of profit, maximizing monopoly. It sells a patented rabies vaccine for pets and earns economic profits.
-
here are 4 common categories of customers: new, existing, exiting, and exited. Suppose you work for a large financial institution that is looking to improve its savings deposits and retirement...
-
Q. Today, social, mobile, and local marketing are the fastest-growing forms of online marketing. This assignment is to plan your own business. A. Describe its business model mentioning the comparison...
-
In 2018, Mr. Colburn created a trust for the benefit of his five adult children and transferred \(\$ 300,000\) to it. The trustee may decide how much income to pay to each child each year. The trust...
-
The nitrogen atoms in N2 participate in multiple bonding, whereas those in hydrazine, N2H4, do not. (a) Draw Lewis structures for both molecules. (b) What is the hybridization of the nitrogen atoms...
-
What are the name and formula of the compound commonly used in the manufacture of glass to increase the index of refraction?
-
Treating oxytocin with certain reducing agents (e.g., sodium in liquid ammonia) brings about a single chemical change that can be reversed by air oxidation. What chemical changes are involved?
-
What classes of reactions are involved in the cleavage of the Fmoc group with piperidine, leading to the unprotected amino acid and the fluorene by-product? Write mechanisms for these reactions.
-
Show all steps in the synthesis of GVA using the tert-butyloxycarbonyl (Boc) group as a protecting group.
-
One Stop Car Wash needs you to create an automated system to keep track of the revenues which are being observed daily. Your assignment is to simulate the traffic through a single car wash booth,...
-
Find the eigenvalues and the corresponding eigenvectors for the following 3x3 matrix. -1 0 A -1 2 0-1
-
(D) What will happen when the following program is compiled and executed? #include int i; void increment( int i) { i++; } int main() { for(i=0; i <10; increment(i)) ( ) printf("%d ", i); return 0; }...

Study smarter with the SolutionInn App


