In the figure, AB CD and BC DE. What is the value of x? A)
Question:
In the figure, A̅B̅ ∥ C̅D̅ and B̅C̅ ∥ D̅E̅. What is the value of x?
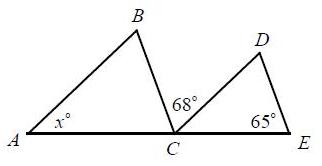
A) 47
B) 51
C) 55
D) 57
Transcribed Image Text:
A B 68° C D 65° E
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
A A B mBCAmDEC mBCA 65 mDCE x 68 C Note Figure no...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Mathematics questions
-
FIGURE EX9.8 is the kinetic-energy graph for a 2.0 kg object moving along the x-axis. Determine the work done on the object during each of the four intervals AB, BC, CD, and DE. K (J) 4- 2 A B C D E...
-
1. How many moles of magnesium is 3.01 x 1022 atoms of magnesium? 2. Find the mass in 2.6 mol of lithium bromide. 3. Use the equation below to answer questions 3a and 3b. C6H12O6 ->2CH5OH + 2CO2 a....
-
if we choose statistic as our keyword, our cipher would be determined as follows: method i. write the word statistic without the repeated letters. then complete the cipher with the unused alphabet...
-
Pierce Phones is considering the introduction of a new model of headphone whose selling price is $18 per unit and whose variable expense is $15 per unit. The company's monthly fixed expense is...
-
Financial information related to Oak Tree Interiors for October and November 2012 is as follows: a. Prepare balance sheets for Oak Tree Interiors as of October 31 and as of November 30, 2012. b....
-
Calculate the pH of a solution that is 1.00 M HCN and 1.00 M HF. Compare the concentration (in molarity) of the CN2 ion in this solution with that in a 1.00 M HCN solution. Comment on the difference.
-
Consider the gold isotope \({ }^{197} \mathrm{Au}\). a. How many electrons, protons, and neutrons are in a neutral \({ }^{197} \mathrm{Au}\) atom? b. The gold nucleus has a diameter of \(14.0...
-
Heritage Furniture Co. uses a standard cost system. One of the companys most popular products is an oak entertainment center that looks like an old icebox but houses a television, stereo, or other...
-
A single proprietorship has a gross receipts of P500,000 from ordinary transactions, capital gains of P120,000 from sale of car and interest income of P4,000, net of final tax of 20%. The optional...
-
The graph depicts a survey of 400 senior students in a high school who took the AP tests last May. The number of AP tests taken by each student ranges from zero to six. By what percent is the number...
-
Effective December 31, 2023, Zintel Corporation proposes to issue additional shares of its common stock in exchange for all the assets and liabilities of Smith Corporation and Platz Corporation,...
-
Provide three considerations when evaluating the sufficiency and appropriateness of audit evidence.
-
Knight Company, a calendar-year rm with 100,000 shares of common stock outstanding at the start of the year, declares a three-for-one stock split halfway through the year. The next day, Knight issues...
-
How can communication contribute to team dynamics? What other factors may contribute to team dynamics?
-
Julio owns 100% of stock of the C corporation The Perm Bank, Inc. (Perm). Perm has total E&P of $20,000 and Julio has a basis of $100,000 in his Perm stock prior to the following distribution. Perm...
-
How can social media retargeting ads be used in the decision stage? a. To reach a new target audience. b. To track post impressions and reach c. To encourage customer interactions and responses d. To...
-
hours and a standard deviation of 0.8 hour. A student is randomly selected. he times per week a student uses a lab computer are normally distributed, with a mean of 5.4 (a) Find the probability that...
-
Find a business plan on the Internet . Examine it to see whether it follows the guidelines provided in this text. Use a highlighter to mark the sections of the plan that are present. Then, make a...
-
Feller Company purchased a site for a limestone quarry for $100,000 on January 2, 2019. It estimate that the quarry will yield 400,000 tons of limestone. It estimates that its retirement obligation...
-
Working for ones self generally means that the amount of time devoted to work is up to the individual. Is there sufficient evidence to conclude that people who work for themselves (WRKSLF: 1 = Self ,...
-
There appear to be many attractive features of government jobs, the most attractive being job security. But is this actually the case? Conduct a test to determine whether government workers (WRKGOVT:...
-
Do men (SEX: 1 = Male, 2 = Female) prefer jobs with higher incomes more than do women? Conduct a statistical test to answer the question. Please look at the list below and specify which one you would...
-
How does Nike mitigate the inherent risks in the international currency?
-
The guidelines are meant to provide additional instruction on assigning and sequencing codes, as well as to assist in identifying certain procedures. The obstetrics guidelines indicate we should...
-
true or false: TO SOURCE DEDUCTIONS, FIRST YOU ALLOCATE TO THE CLASS OR CLASSES OF GROSS INCOME THAT APPLIES TO THAT EXPENSE. THEN YOU APPORTION TO THE VARIOUS COUNTRIES THAT APPLIES TO THE INCOME...

Study smarter with the SolutionInn App


